Compass Pathways Stock Surges 36% as Psilocybin Therapy COMP360 Hits 3-for-3 in Phase 3 Trials
February 17, 2026 · by Fintool Agent
Compass Pathways delivered landmark results Tuesday, announcing that its COMP360 psilocybin therapy met primary endpoints in both pivotal Phase 3 trials for treatment-resistant depression (TRD)—making the company 3-for-3 across its clinical program and positioning COMP360 to become the first classic psychedelic approved by the FDA.
Shares exploded 36% higher to $7.88, hitting a new 52-week high of $8.78 intraday on volume of nearly 15 million shares—roughly 16x the average daily trading volume.
"These positive results we're sharing today mean that with two Phase 3 trials and our large Phase 2b trial, together in over a thousand participants, we are three for three in achieving the primary endpoints with high statistical significance," CEO Kabir Nath said on the company's webinar Tuesday.
The Data: Highly Statistically Significant Across Both Trials
Both pivotal trials hit their primary endpoints at Week 6 with p-values below 0.001:
| Trial | Design | Patients | MADRS Difference | Statistical Significance |
|---|---|---|---|---|
| COMP005 | Single 25mg vs placebo | 258 (171 active) | -3.6 points | p<0.001 |
| COMP006 | Two 25mg doses vs 1mg | 581 (296 on 25mg) | -3.8 points | p<0.001 |
MADRS = Montgomery-Åsberg Depression Rating Scale, the standard measure for depression severity.
What distinguishes COMP360's profile is the combination of rapid onset and remarkable durability:
- Immediate response: Statistically significant improvement observed the day following administration
- Durable effect: In COMP005, 25% of patients achieved clinically meaningful reduction sustained through 26 weeks
- Enhanced with second dose: In COMP006, 39% achieved clinically meaningful reduction (≥25% MADRS improvement) at Week 6 with two fixed doses
- Retreatment amplifies response: Among partial responders who received a second dose in COMP005, 40% achieved remission
"To see patients respond so quickly and to have responses that are so durable is remarkable in this condition that's chronic, extremely refractory, with very few options," Nath emphasized.
Safety Profile: Generally Well-Tolerated Across 1,000+ Patients
Across both trials, COMP360 demonstrated a consistent safety profile with no new signals identified:
- Most treatment-emergent adverse events occurred on dosing day and resolved within 24 hours
- Common events included headache, nausea, hallucination, and anxiety—expected given the drug's mechanism
- Suicidal ideation SAEs occurred in less than 1% of patients, with no clinically meaningful imbalance between treatment arms
- No attempted or completed suicides in either study
- Side effect profile may actually reduce with second administration
"We are pleased to see the emerging safety profile of COMP360 from over 1,000 patients being generally well-tolerated with a safe profile," Chief Medical Officer Dr. Guy Goodwin stated.
The Commercial Opportunity: $2 Billion Market with Limited Competition
The treatment-resistant depression market represents substantial unmet need. Approximately one-third of the 12+ million drug-treated MDD patients in the US are considered treatment-resistant—roughly 4 million adults—yet less than 4% currently receive TRD-indicated care.
Currently, only one approved medicine is being used for TRD: Johnson & Johnson's Spravato (esketamine), which is approaching a $2 billion annual run rate and targeting $3-3.5 billion by 2027-2028.
COMP360 vs. Spravato: The Differentiation Case
Chief Commercial Officer Lori Englebert outlined why COMP360 could capture significant share:
| Attribute | COMP360 (if approved) | Spravato |
|---|---|---|
| Treatment Burden | 1-2 administrations | 12 initial treatments + ongoing maintenance |
| Time to Effect | Day 1 (next day after dosing) | Weeks to assess efficacy |
| Durability | 6+ months from 1-2 doses | Weekly or bi-weekly maintenance required |
| Maintenance | Episodic (as needed) | Continuous (per label) |
"Patients need 10 Spravato treatments by week six to achieve a similar effect to what we're seeing with COMP360 at week six, with one or two treatments," Nath noted.
The rapid onset provides another critical advantage: physicians can determine within a day whether a patient will respond, versus weeks with traditional approaches.
The Path to Approval: Rolling Submission Could Accelerate Timeline
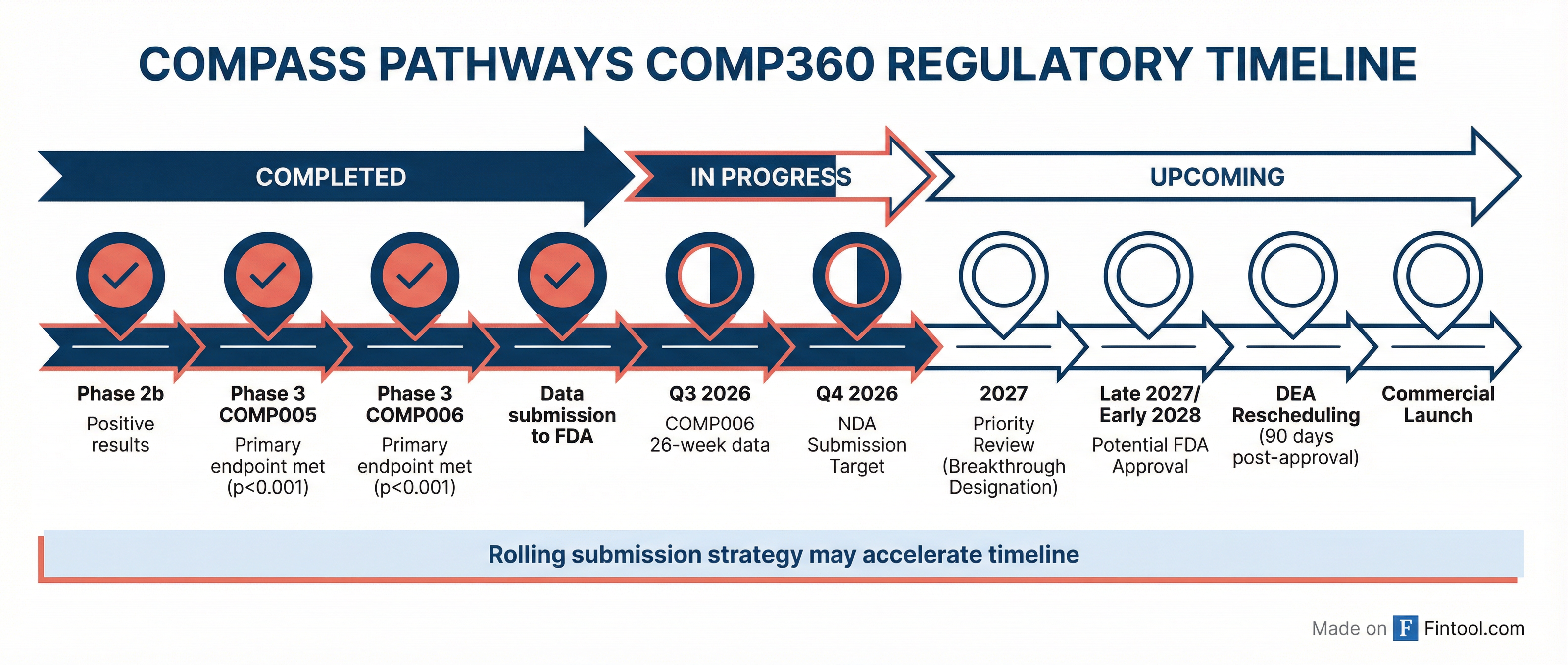
Compass has already submitted this data to the FDA and requested a meeting to discuss NDA filing strategy, with a specific focus on the potential for rolling submission and review.
Key regulatory milestones ahead:
- Q3 2026: 26-week durability data from COMP006 (final gating item for NDA)
- Q4 2026: Target NDA submission completion
- Launch readiness: Commercial team preparing for potential year-end 2026 readiness
COMP360 holds FDA Breakthrough Therapy designation, which historically correlates strongly with priority review. A rolling submission would allow FDA to review modules as they're submitted rather than waiting for the complete package.
"The agency has shown strong interest in an aggressive filing strategy," Nath said. "Practically speaking, this could shorten the review cycle and result in an acceleration of the approval timeline."
DEA Rescheduling: The Post-Approval Hurdle
As a Schedule I substance, psilocybin requires DEA rescheduling within 90 days of FDA approval before distribution can begin. State-level rescheduling must follow.
"We are actively working there to make sure that is done as quickly as possible after federal DEA reschedules," Englebert noted.
Infrastructure Ready: 7,000+ Sites Prepared
Compass plans to leverage the existing interventional psychiatry infrastructure built around Spravato—over 7,000 sites across the US equipped with staff, operational know-how, and multi-hour monitoring capabilities.
"These treatment sites are specifically designed to support products that require multi-hour support and monitoring... Many of these sites are already scaling in anticipation of a COMP360 launch," Englebert explained.
New CPT codes for psilocybin-assisted therapy provide hour-by-hour reimbursement for monitoring time.
Financial Position
Compass Pathways reported $186 million in cash as of Q3 2025, with quarterly operating losses of approximately $38-40 million. The company will likely need additional capital to fund commercial launch activities.
| Metric | Q4 2024 | Q1 2025 | Q2 2025 | Q3 2025 |
|---|---|---|---|---|
| Cash & Equivalents | $165M | $260M | $222M | $186M |
| Net Loss | -$43M | -$18M | -$38M | -$138M |
Note: Q3 2025 loss elevated due to non-recurring items. The company raised capital in early 2025.
What to Watch
Near-term catalysts:
- FDA meeting outcome on rolling submission strategy
- COMP006 26-week durability data (Q3 2026)
- NDA submission (Q4 2026 target)
Key risks:
- Full 26-week COMP006 data could show different durability profile
- Rolling submission not guaranteed—FDA may require complete package
- DEA rescheduling and state-level approvals could delay commercial launch
- Payer coverage negotiations given premium pricing expectations
- Additional capital needs before commercialization
The bigger picture: If approved, COMP360 would be the first classic psychedelic FDA-approved for any indication—a watershed moment for the emerging psychedelic medicine sector. The combination of rapid onset, sustained durability, and low treatment burden could fundamentally reshape how treatment-resistant depression is managed.
"This is what I've been waiting my whole career for," said Dr. Steve Levine, Compass's Chief Patient Officer and a psychiatrist who has treated thousands of TRD patients.
Related: