Disc Medicine Defends Path Forward After FDA Rejects Bitopertin in Rare Disease Setback
February 17, 2026 · by Fintool Agent

Disc Medicine hosted an investor call Tuesday morning to address the fallout from the FDA's unexpected rejection of bitopertin, the company's lead drug for erythropoietic protoporphyria (EPP)—and the first experimental therapy to be rejected after going through the new Commissioner's National Priority Voucher program.
The stock fell as much as 44% on Friday following news of the Complete Response Letter before recovering to close down 22% at $55.95. Shares remain under pressure, erasing most of the gains from the company's JPM Healthcare Conference presentation in January when investors were positioning for an imminent approval.
CEO Quisel: "Readily Addressable"
On Tuesday's call, CEO John Quisel struck a confident tone despite the setback, calling the FDA's concerns "readily addressable" with results from the ongoing Phase 3 APOLLO trial.
"We wish we were here today discussing better news," Quisel acknowledged before pivoting to offense. "Our commitment to bringing this program forward and pursuing an approval has not wavered. We believe the issue raised in this Complete Response Letter is readily addressable with the results of the ongoing Phase 3 APOLLO study."
The key issue: while the FDA acknowledged that bitopertin significantly lowers protoporphyrin IX (PPIX)—the toxic metabolite that causes EPP patients to experience severe pain when exposed to sunlight—the agency questioned whether this biomarker reduction was "reasonably likely to predict clinical benefit."
The FDA specifically called the 40% PPIX reduction seen in Disc's Phase 2 trials "relatively modest" and noted the lack of statistical correlation between biomarker reduction and improved sunlight tolerance.
The APOLLO Path Forward
Management emphasized that the Phase 3 APOLLO trial is well-positioned to provide the clinical evidence the FDA requires. The trial is enrolling 150 patients with EPP and uses two co-primary endpoints that directly address the FDA's concerns:
- PPIX Reduction: Measuring percent change in whole-blood metal-free protoporphyrin IX
- Time in Sunlight Without Pain: A clinical endpoint measuring patients' ability to tolerate light exposure during month six
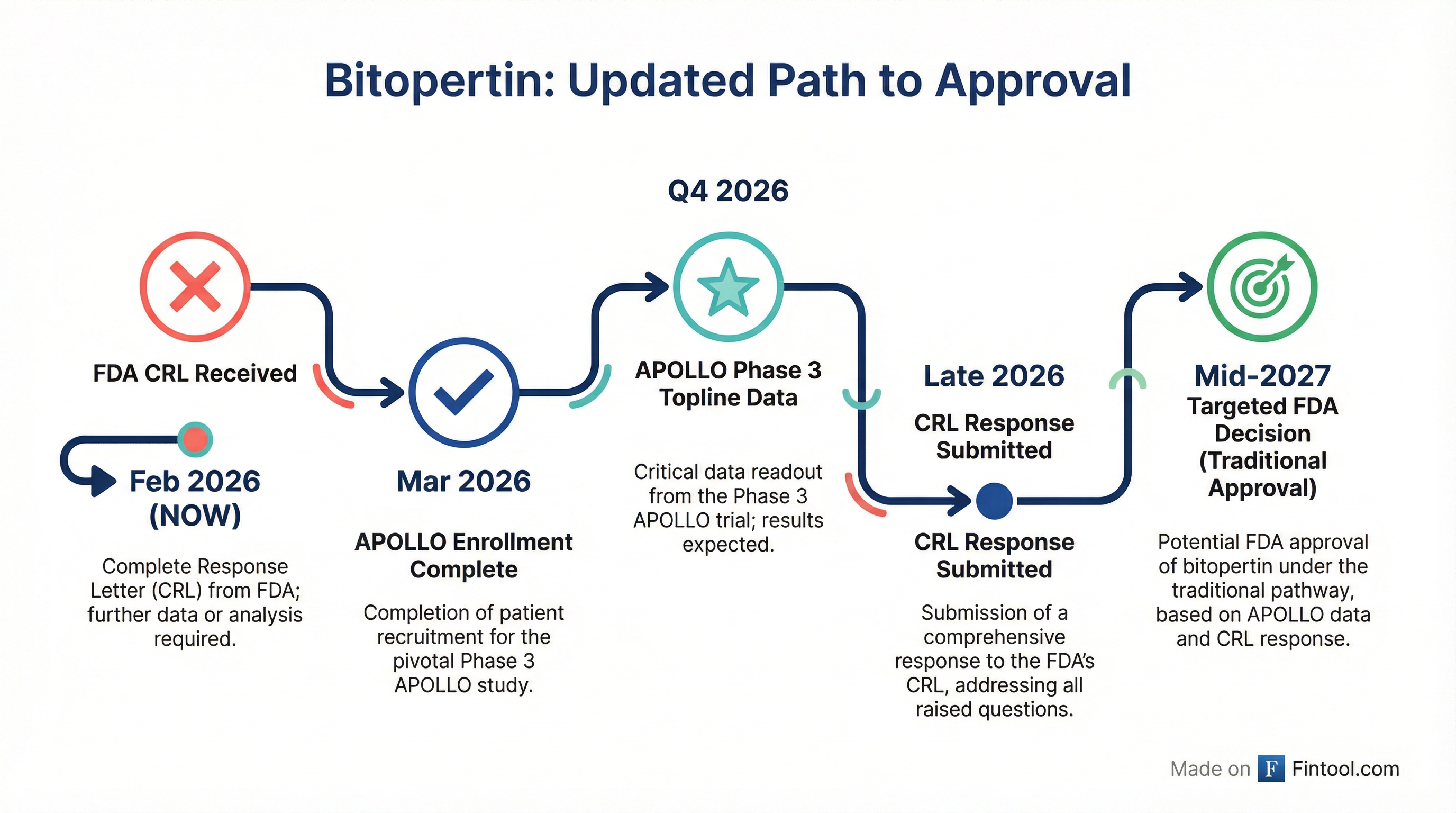
Critically, Quisel announced that APOLLO enrollment will complete in March 2026—several months earlier than planned due to "overwhelming enthusiasm" from the EPP patient community. A blinded sample size recalculation confirmed no need to increase enrollment, suggesting the trial is tracking well against its statistical assumptions.
"We have a lot of confidence in the design and chosen endpoint," Quisel said. "Both co-primaries need to hit p-value of 0.05."
| Milestone | Timeline |
|---|---|
| APOLLO Enrollment Complete | March 2026 |
| APOLLO Topline Data | Q4 2026 |
| CRL Response Submission | Late 2026 |
| FDA Decision (Traditional Approval) | Mid-2027 |
No Surprises Expected in Type A Meeting
Several analysts pressed management on whether the FDA might change its position on APOLLO's endpoints. Quisel was emphatic that the company sees "no evidence" of any misalignment:
"The CRL is essentially pointing to the APOLLO trial as designed as being the path to getting the dataset that they would view as appropriate for supporting approval. We feel good about the way the APOLLO trial is designed and the degree to which it's been vetted with the FDA."
The company plans to request a Type A meeting to confirm its approach, with minutes typically available within 30 days.
First Test of Makary's FDA
The rejection marks the first experimental drug to be turned down after going through FDA Commissioner Marty Makary's new Commissioner's National Priority Voucher (CNPV) program, which promised expedited reviews for companies aligned with "U.S. national priorities."
Reports circulated in January that FDA biologics head Vinay Prasad was skeptical of bitopertin's efficacy—concerns that appear to have been validated. Truist Securities analysts called the rejection "surprising" given bitopertin met "all key criteria for accelerated approval" in their view.
"It appears that recent media reports regarding Vinay Prasad's skepticism of IRON's bitopertin were well-founded," Truist wrote Friday.
The episode highlights a broader tension in FDA policy around accelerated approvals based on surrogate endpoints—a debate that has intensified under the current administration.
Pipeline Provides Cushion
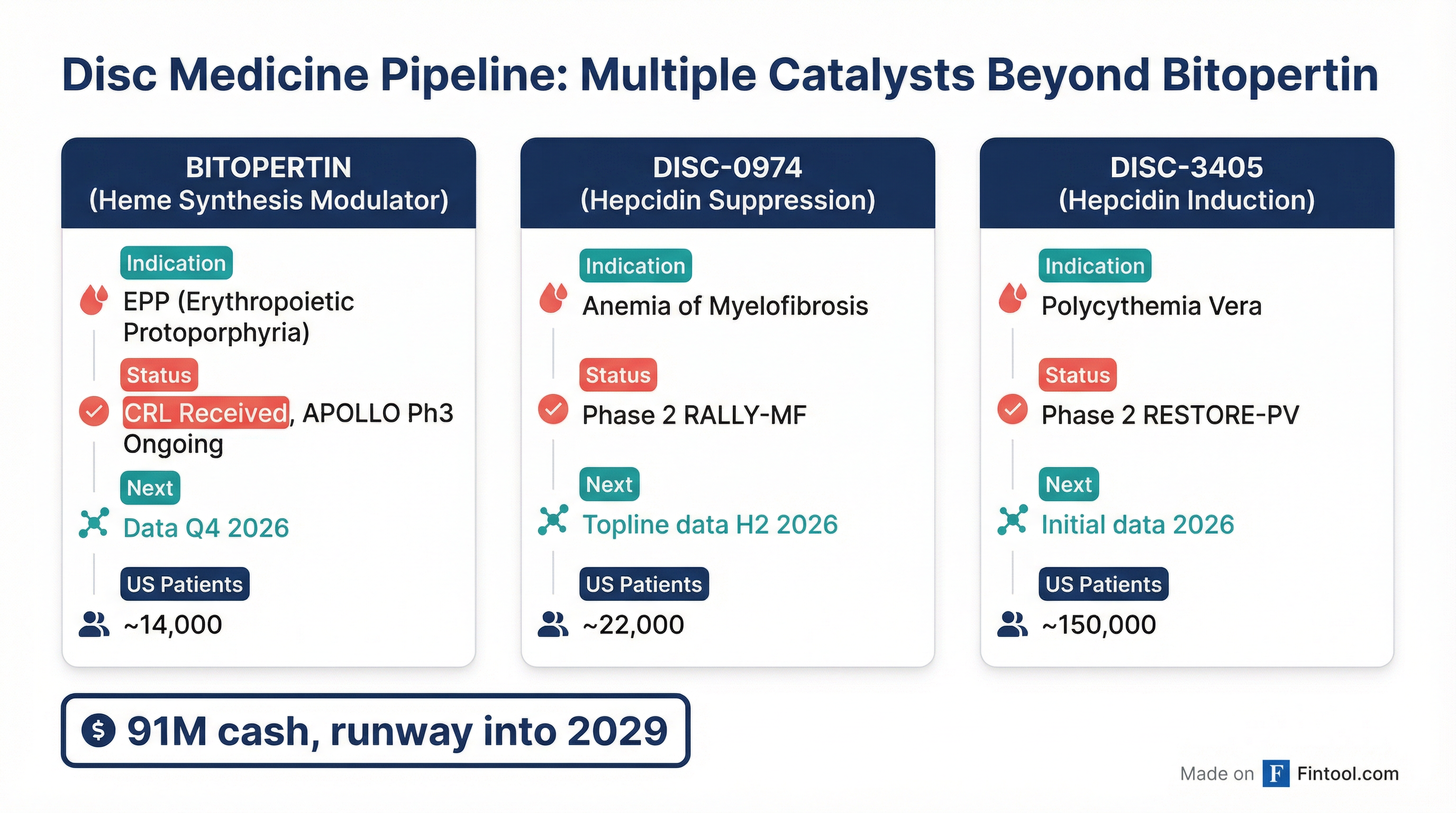
While bitopertin's delay is significant, Disc Medicine emphasized its diversified pipeline continues to advance. The company has three clinical-stage programs targeting red blood cell disorders across different mechanisms:
| Program | Indication | Status | Next Catalyst |
|---|---|---|---|
| Bitopertin | EPP (14K US patients) | Phase 3 APOLLO | Topline Q4 2026 |
| DISC-0974 | Anemia of Myelofibrosis (22K) | Phase 2 RALLY-MF | Topline H2 2026 |
| DISC-3405 | Polycythemia Vera (150K) | Phase 2 RESTORE-PV | Initial data 2026 |
| DISC-3405 | Sickle Cell Disease (1M+) | Phase 1b | Initial data H2 2026 |
"As a company, we are in great shape here to deliver across a wide range of programs," Quisel noted, pointing to multiple data readouts expected throughout 2026.
Financial Position Remains Strong
The company reiterated its cash guidance, with approximately $791 million in cash, cash equivalents, and marketable securities as of December 31, 2025—providing runway into 2029.
This cushion is critical given the 18-month delay to potential bitopertin approval and the need to fund both the APOLLO trial completion and ongoing Phase 2 studies for DISC-0974 and DISC-3405.
Management indicated it would reevaluate commercial spending plans given the extended timeline. Pre-launch activities like physician account validation will continue, but broader commercial build-out decisions are under review.
What to Watch
Near-term catalysts:
- Type A meeting with FDA (timing TBD) to confirm APOLLO alignment
- APOLLO enrollment completion in March 2026
- DISC-0974 RALLY-MF Phase 2 topline data (H2 2026)
Key risks:
- APOLLO trial execution risk—both co-primaries must achieve p<0.05
- Further FDA guidance shifts under the Makary administration
- Commercial organization timing and spend optimization
The path to approval is now longer but arguably cleaner. A traditional approval based on clinical endpoints would remove the overhang of post-marketing confirmatory study requirements that come with accelerated approval—and potentially support stronger pricing discussions with payers.
For EPP patients who have participated in clinical trials and experienced life-changing relief from a debilitating disease, the 18-month wait feels less like a technicality and more like a tragedy. As one analyst noted, the patient stories of "beach vacations with family" and "spending time on boats for the first time" underscore what's at stake.
Related
Sources: Disc Medicine investor call (February 17, 2026), company press releases, FDA Complete Response Letter, analyst reports