Gilead Pays $7.8B to Take Full Control of CAR-T Challenger to J&J's Carvykti
February 23, 2026 · by Fintool Agent

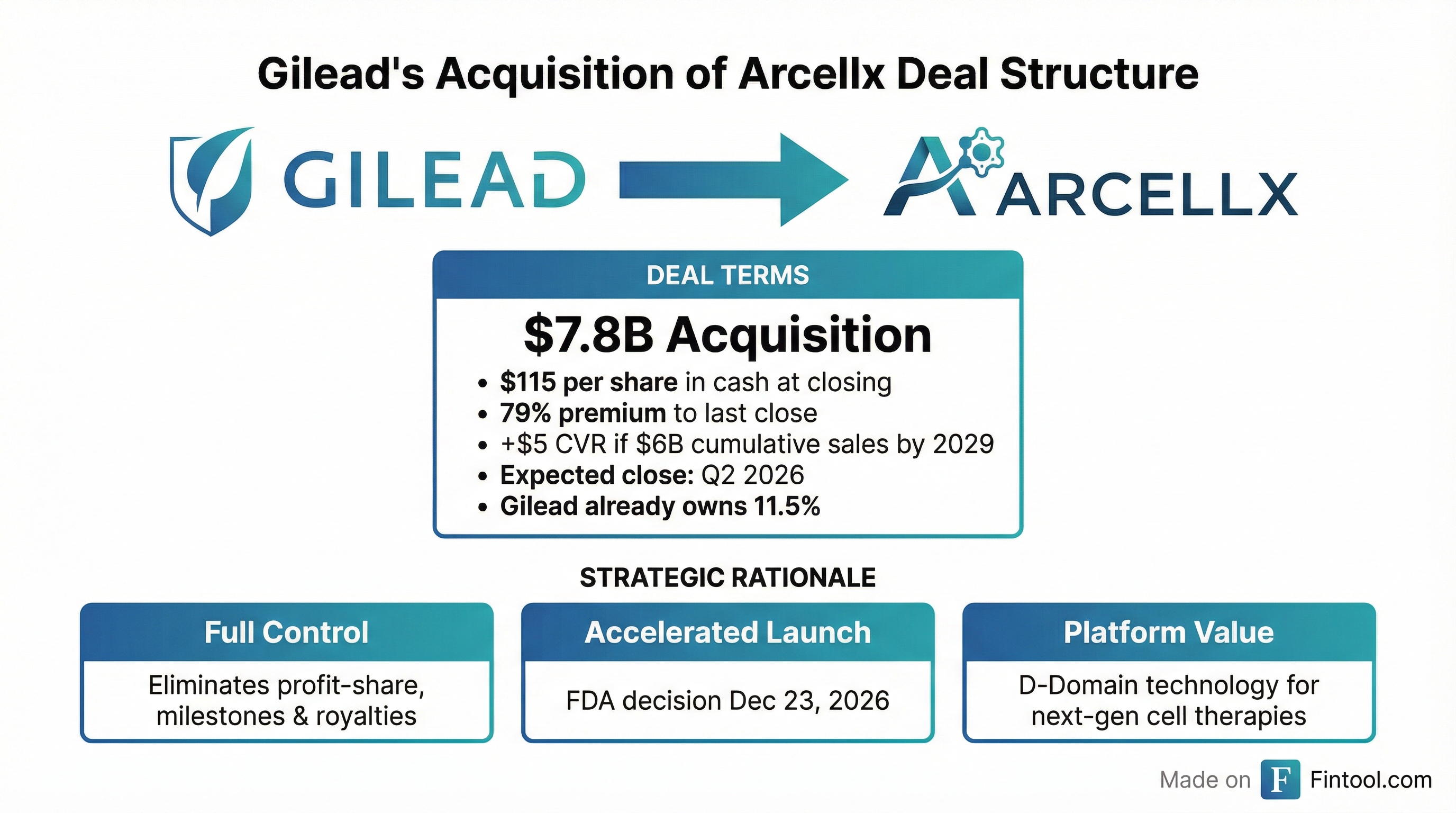
Gilead Sciences is acquiring Arcellx for $7.8 billion in cash to take full control of a CAR-T cell therapy that's poised to challenge Johnson & Johnson and Legend Biotech's Carvykti in the blockbuster multiple myeloma market.
The deal comes just days after CEO Daniel O'Day told investors M&A was "not an urgent priority"—and with an FDA decision on the therapy expected in ten months.
Arcellx shares surged 78% to $114.46 in premarket trading, while Gilead fell about 1%.
The Deal
Gilead is paying $115 per share in cash—a 79% premium to Arcellx's Friday close—plus a contingent value right worth $5 per share if cumulative anito-cel global sales reach $6 billion from launch through the end of 2029.
The acquisition gives Gilead full control of anito-cel, eliminating the profit-share, milestones, and royalties it would have owed under the existing partnership. Gilead already owns approximately 11.5% of Arcellx's outstanding shares.
"This agreement reflects our conviction in the potential of anito-cel and our intention to move with speed so we can make the most of that potential for patients with multiple myeloma," said Daniel O'Day, Chairman and CEO of Gilead Sciences. "Beyond the potential launch this year, anito-cel could become a foundational treatment for multiple myeloma over time, including earlier lines of therapy."
From Partner to Target: The Three-Year Journey
The acquisition represents the culmination of a partnership that began in December 2022. Here's how the relationship evolved:
| Date | Event | Value |
|---|---|---|
| Dec 2022 | Initial partnership with Kite | $225M upfront + $100M equity + up to $3.9B milestones |
| Nov 2023 | Expanded collaboration | $85M cash + $200M equity + additional milestones |
| May 2025 | iMMagine-1 pivotal results | 97% ORR, 68% CR/sCR reported |
| Feb 2026 | Full acquisition | $7.8B ($115/share + $5 CVR) |
Under the original deal, Gilead was on the hook for $530 million in remaining milestones for anito-cel alone. The acquisition effectively converts those potential payments—plus profit-sharing and royalties—into a single upfront transaction.
Why Now: The Clinical Case for Anito-cel
The FDA accepted Arcellx's BLA for anito-cel as a fourth-line treatment for relapsed or refractory multiple myeloma, with a PDUFA action date of December 23, 2026.
The filing was supported by compelling data from the pivotal Phase 2 iMMagine-1 study:
| Metric | Result |
|---|---|
| Overall Response Rate | 97% |
| Complete Response / Stringent CR | 68% |
| 6-Month Progression-Free Survival | 91.9% |
| 6-Month Overall Survival | 96.6% |
More importantly for commercial positioning, Guggenheim Securities analysts recently noted that physicians treating multiple myeloma patients prefer anito-cel to the incumbent Carvykti based on safety data.
"We really feel strongly about the differentiated profile of anito-cel and see it not just as a best-in-class therapy, but the best option for fourth line plus myeloma patients based on both the efficacy data and the safety data," Gilead management said at a recent investor conference.
The Competitive Battlefield
Anito-cel enters a market dominated by J&J and Legend Biotech's Carvykti, which has established itself as the fastest-launching CAR-T therapy in history.

Legend Biotech CEO Ying Huang outlined Carvykti's position at the J.P. Morgan Healthcare Conference in January: "Carvykti has been the fastest launch among CAR-T therapies today and has generated about $1.7 billion in net trade sales in the last 12 months through the third quarter of 2025."
The J&J/Legend partnership targets more than $5 billion in peak annual sales and has already treated over 10,000 patients. Carvykti is offered at 279 global treatment sites across 14 countries.
But Gilead has a significant infrastructure advantage: Kite currently operates 177 authorized treatment centers in the U.S. from its existing leukemia business. Management's goal is to have the majority of those centers onboarded for anito-cel within 30 days of launch.
Kite Needs a Win
The acquisition comes at a critical time for Gilead's cell therapy unit. Kite's existing portfolio—Yescarta and Tecartus for leukemia and lymphoma—has been losing ground to competitors.
| Metric | FY 2025 | FY 2024 | Change |
|---|---|---|---|
| Cell Therapy Revenue | $1.8B | $1.9B | -7% |
| 2026 Guidance | $1.6B | — | -10% YoY expected |
"For 2026, we continue to expect these competitive headwinds, including in several countries outside the U.S., where we expect new entrants this year. Overall, we expect Kite revenue to decline approximately 10% in 2026 compared to 2025," Gilead's Chief Commercial Officer Johanna Mercier said on the Q4 earnings call.
The multiple myeloma market represents a significant new opportunity. Gilead pegs the fourth-line-plus market alone at approximately $3.5 billion.
Financial Firepower
Gilead has the balance sheet capacity to absorb the deal:
| Gilead Financials | FY 2025 | FY 2024 | FY 2023 |
|---|---|---|---|
| Revenue | $29.4B | $28.8B | $27.1B |
| Net Income | $8.5B | $0.5B | $5.7B |
| Cash & Equivalents | $7.6B | $10.0B | $6.1B |
| Total Debt | $24.6B | $27.3B | $25.7B |
The deal is expected to be accretive to earnings per share in 2028 and beyond, assuming FDA approval of anito-cel.
Platform Upside
Beyond anito-cel, Gilead is acquiring Arcellx's D-Domain CAR technology platform, which has generated proprietary target-binding domains with improved specificity and enhanced binding affinity.
"In addition, the anito-cel D-domain BCMA binder could be important to our work in in vivo cell therapy, further strengthening our potential in oncology and inflammation," O'Day said.
Gilead has been building its in vivo cell therapy capabilities through recent partnerships with Interius and Pregene, betting that off-the-shelf therapies could eventually replace the complex autologous manufacturing process that requires using each patient's own cells.
What to Watch
December 23, 2026: FDA PDUFA action date for anito-cel in fourth-line multiple myeloma
Q2 2026: Expected deal closing, subject to Arcellx shareholder tender, regulatory approvals, and customary conditions
2028: Year the deal is expected to become EPS accretive
$6 billion by 2029: Cumulative sales threshold that triggers the $5 CVR payment