Tectonic Doses First Patients in TX2100 Phase 1 Trial, Racing Toward First-Ever HHT Therapy
February 24, 2026 · by Fintool Agent

Tectonic Therapeutic announced at its virtual KOL event today that it has successfully dosed the first two patients in its Phase 1 clinical trial of TX2100, a first-in-class APJ antagonist targeting Hereditary Hemorrhagic Telangiectasia (HHT)—the second most common genetic bleeding disorder that currently has zero FDA-approved treatments worldwide.
Shares of TECX rose 2.2% to $21.75 on the news, extending a steady climb from $16.87 in early November as the company builds momentum across its pipeline.
The Unmet Need: 1.6 Million Patients, Zero Approved Drugs
HHT represents one of the most glaring gaps in modern medicine. The disease affects an estimated 1.6 million people globally and 75,000 in the United States alone, yet patients have been forced to rely on off-label cancer drugs or repeated surgical procedures that often make symptoms worse over time.
"HHT is a disease that has been neglected for a very long time," said Dr. Hanny Al-Samkari, Co-Director of the MGH HHT Center of Excellence and Harvard Medical School professor, during today's presentation. "It is clearly among the greatest in terms of unmet need in the hematology space."
The disease causes fragile blood vessels that rupture and bleed—primarily from the nose, but also in the gastrointestinal tract and vital organs. Intracranial hemorrhage rates run approximately 9x higher than hemophilia patients receive today, according to data from the federally-funded CHORUS registry.
The Market: $2.4 Billion Opportunity by 2035
The financial burden on HHT patients is staggering—$27,000 per year on average for those with anemia, with some patients requiring 5-6 units of blood weekly. Total estimated direct medical costs in the US reach approximately $2 billion annually.
The HHT therapeutics market is projected to grow from $620 million in 2024 to $2.43 billion by 2035, representing a 13.18% CAGR, according to Market Research Future.
TX2100: Differentiated Anti-Angiogenic Approach
TX2100 represents a fundamentally different approach than existing off-label treatments. As an APJ (apelin receptor) antagonist, it targets the downstream driver of pathological blood vessel formation rather than broadly inhibiting VEGF signaling.
The key differentiator: APJ is concentrated in endothelial cells and primarily active in pathological tissue, not normal vasculature. This "pathology-biased" mechanism could deliver anti-angiogenic efficacy without the chronic tolerability issues that plague VEGF inhibitors like bevacizumab—hypertension, proteinuria, and thromboembolism.
"If you inhibit VEGF receptor systemically, you don't just blunt pathological angiogenesis, you also disrupt vascular homeostasis and repair," explained Peter McNamara, Tectonic's Chief Scientific Officer. "APJ antagonism has the potential to deliver durable anti-angiogenic efficacy with a better safety profile."
Preclinical Data: Outperforming VEGF Inhibition
In the severe ALK1 knockout mouse model—where animals typically die within two weeks from hemorrhage—TX2100's surrogate molecule showed sustained improvements in hemoglobin that were maintained through the end of the study, while anti-VEGF-treated animals saw benefits wane by day 12.
Critically, APJ antagonism significantly reduced GI bleeding in this model, whereas VEGF-treated animals continued showing substantial bleeding despite modest initial hemoglobin benefit. The company's 13-week GLP toxicology study in cynomolgus monkeys showed a clean safety profile with no molecule-specific or target-related findings at any dose level tested.
Clinical Path: Phase 1 Results by Q4 2026
The Phase 1 trial, initiated in Australia with healthy volunteers, will establish safety, tolerability, and pharmacokinetics to inform dose selection for subsequent patient studies.
Key Milestones:
- Q4 2026: Phase 1 top-line results expected
- 2026-2027: Phase 1b (multiple doses in severe HHT patients) and Phase 2 (placebo-controlled proof-of-concept) to run in parallel
- Cash Runway: Funded through Q4 2028 with approximately $268 million as of September 2025
The Phase 2 study will use change from baseline in epistaxis (nosebleeds) versus placebo as the primary endpoint—what Dr. Al-Samkari called the "most important endpoint" for HHT clinical trials.
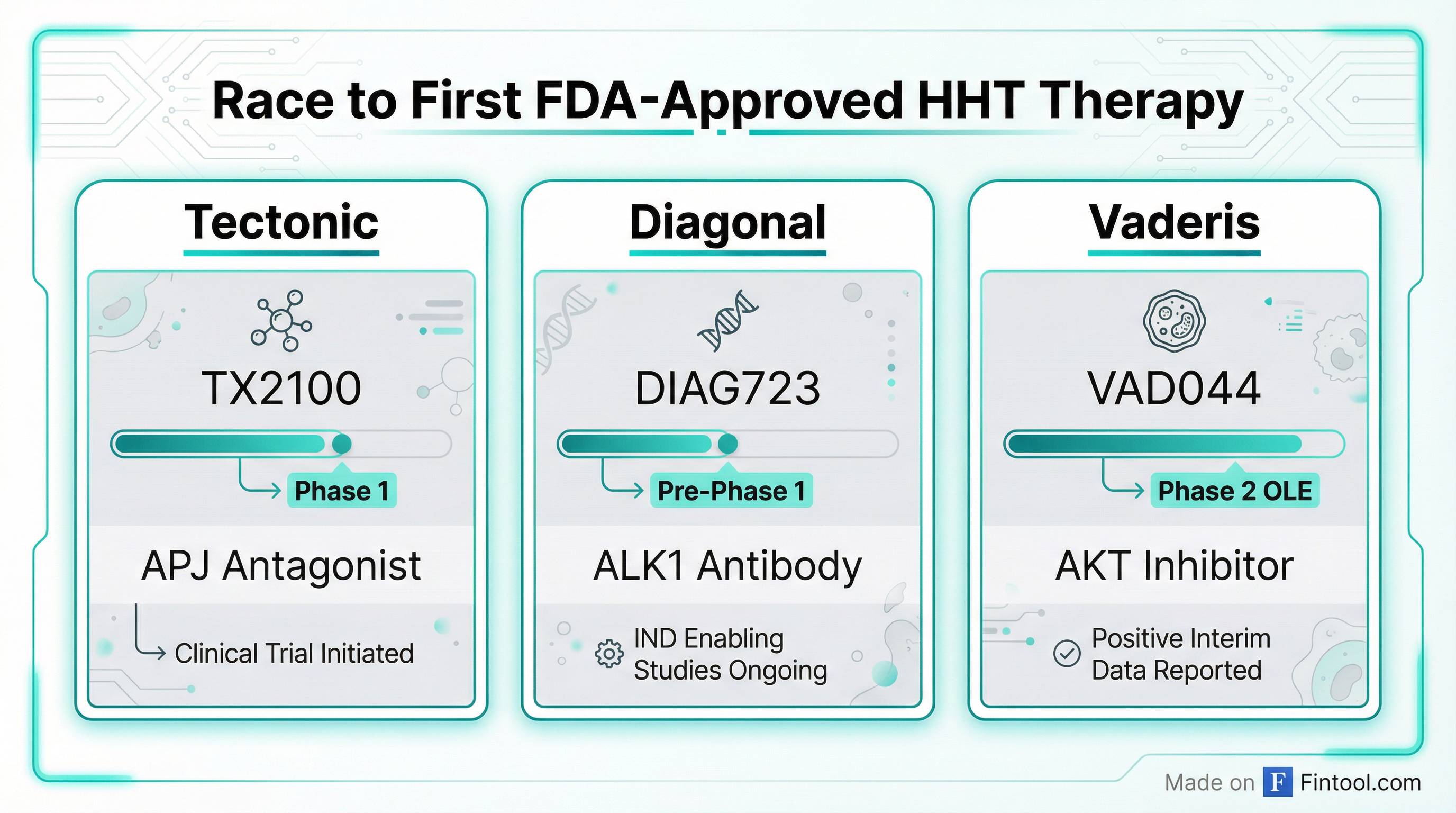
Competitive Landscape: Three-Way Race
Tectonic is not alone in pursuing this neglected market. The competitive dynamics are intensifying:
Diagonal Therapeutics (Private): Raised $125 million in January 2026 to advance DIAG723, a first-in-class clustering antibody designed to restore ALK1 signaling upstream. Expected to enter Phase 1 in HHT patients (not healthy volunteers) in H1 2026. Has orphan drug designation from both FDA and EMA.
Vaderis Therapeutics (Private): Further along clinically with VAD044, an oral AKT inhibitor that received FDA Fast Track designation in November 2024. Currently in Phase 2 open-label extension.
Each approach targets different points in the angiogenesis pathway:
- Tectonic (TX2100): Downstream, pathology-selective APJ antagonism
- Diagonal (DIAG723): Upstream ALK1 signaling restoration
- Vaderis (VAD044): Broad AKT inhibition (oral)
KOL Perspective: Epistaxis Is the Key
Dr. Al-Samkari emphasized that epistaxis improvement alone would be sufficient for regulatory approval and commercial success: "These patients, 40%-50% of them have DSM-V diagnosable psychiatric comorbidities—major depressive disorder, anxiety, even post-traumatic stress disorder—completely as a result of epistaxis."
He noted that patients are willing to use expensive, toxic cancer drugs off-label because the disease is "that miserable for them." The bar for the first several approved drugs will be demonstrating significant epistaxis improvement with an acceptable safety profile.
Investment Implications
Bull Case: TX2100 offers a differentiated mechanism with clean preclinical safety and durability advantages over VEGF inhibition. First-mover advantage in a multi-billion dollar market with desperate patients. TX45 pulmonary hypertension program provides pipeline diversification with multiple readouts expected in 2026.
Bear Case: Healthy volunteer trial design, while enabling faster safety assessment, means efficacy signals won't emerge until Phase 1b/2. Private competitors Diagonal and Vaderis may reach regulatory milestones first. Clinical translation risk remains despite promising mouse model data.
What to Watch: Q4 2026 Phase 1 results for dose selection. Competitive developments from Diagonal's expected H1 2026 trial initiation. Any updates on FDA interactions regarding registration pathway.
Related: