ANAVEX LIFE SCIENCES (AVXL)·Q1 2026 Earnings Summary
Anavex Beats EPS by 60%, Cash Surges to $132M as Burn Rate Drops Sharply
February 9, 2026 · by Fintool AI Agent
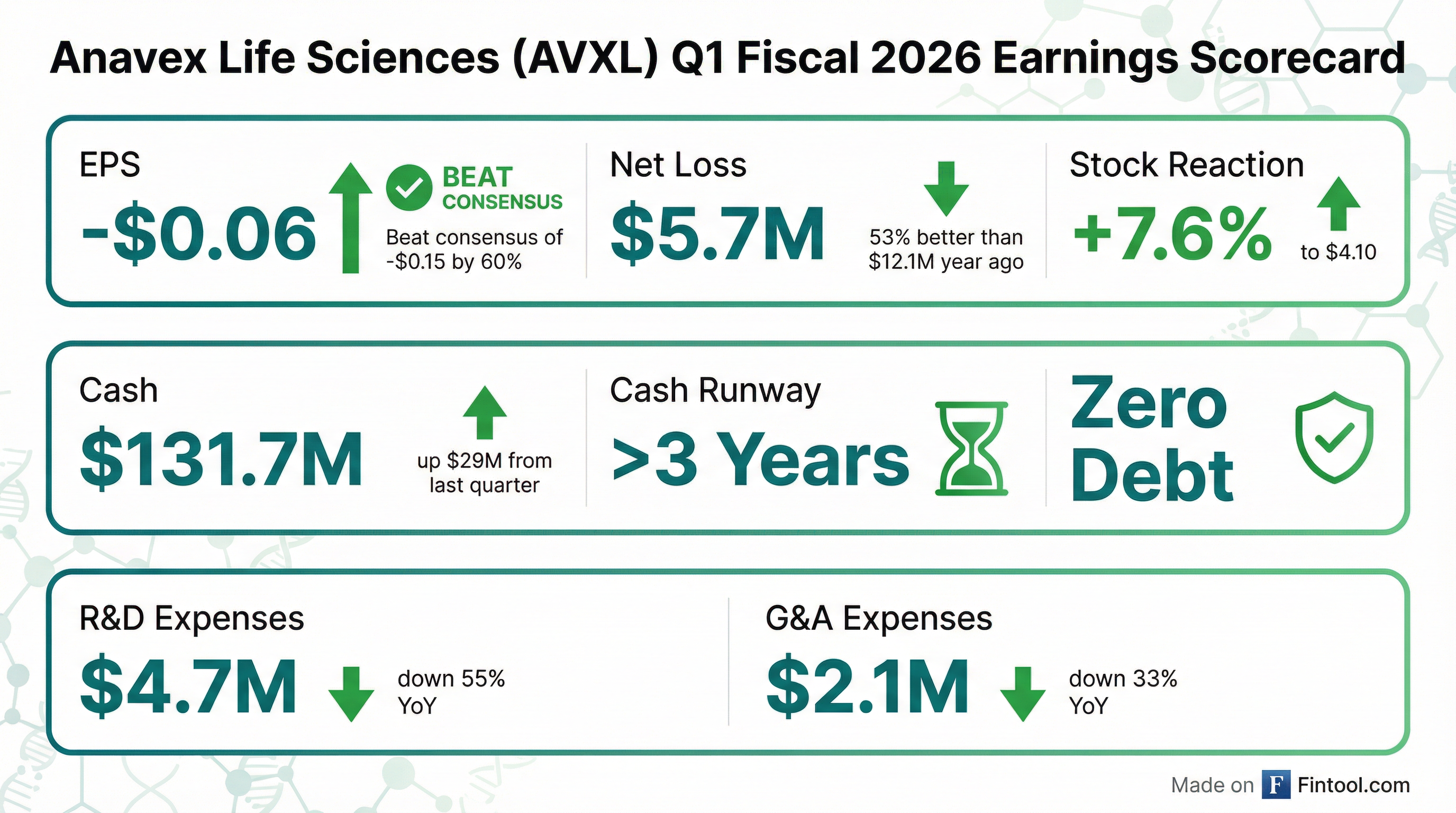
Anavex Life Sciences (NASDAQ: AVXL) delivered a significant earnings beat in Q1 FY2026, reporting EPS of -$0.06 versus consensus expectations of -$0.15—a 60% beat. The clinical-stage biotech, focused on Alzheimer's disease and other CNS disorders, saw its cash position surge to $131.7M while slashing operating expenses by more than 50% year-over-year.
Shares rose 7.6% to $4.10 in regular trading, with aftermarket trades pushing to $4.30.
Did Anavex Beat Earnings?
Yes, and by a wide margin. The quarter marked a significant improvement across all financial metrics:
The EPS beat versus consensus (-$0.06 actual vs. -$0.15 expected) reflects the company's sharply reduced burn rate as clinical programs transition from active enrollment phases.*
*Values retrieved from S&P Global
What Changed From Last Quarter?
Cash Position Strengthened Significantly
The most notable change was the $29M increase in cash, from $102.6M to $131.7M. This appears to be driven by an equity raise during the quarter, evidenced by the increase in additional paid-in capital from $477.2M to $514.7M.
Management emphasized the strengthened balance sheet: "The Company anticipates at its current cash utilization rate, an approximate cash runway of more than 3 years."
Operating Expenses Cut in Half
R&D spending dropped 55% YoY as the company completed the enrollment phase of its Phase IIb/III Alzheimer's trial. G&A expenses were trimmed 33%, demonstrating disciplined cost management.
Principal Financial Officer Sandra Boenisch attributed the decrease to "the completion of a large manufacturing campaign of blarcamesine conducted in Fiscal 2025, and a decrease in clinical trial activities as a result of the completion of our Anavex 3-71 Phase II study in schizophrenia."
The company utilized $7.1 million in operating activities during the quarter after adjusting for non-cash working capital changes.
What Did Management Say About Regulatory Pathways?
FDA Engagement – Constructive
On January 6, 2026, Anavex announced feedback from an FDA Type C meeting where "the FDA shared their interest and collaborative approach to Anavex' development plans." The meeting discussed potential pathways to support a New Drug Application (NDA) for blarcamesine in Alzheimer's disease. Anavex will submit existing data from its Phase IIb/III program as requested by the Agency.
EU – Setback, but Appealing
In December 2025, the European CHMP adopted a negative opinion on the marketing authorization application for blarcamesine. Anavex has requested the EMA to re-examine its opinion, and the appeal process will be led by a different rapporteur and co-rapporteur.
CEO Christopher Missling stated: "We look forward to working with the regulatory agencies in Europe and the U.S. to advance oral blarcamesine as a potential new treatment option for patients."
Q&A Highlights
EMA Re-Examination Timeline and Process
Analysts pressed management on the EMA appeal process. CEO Missling confirmed the re-examination follows a 60+60 day structure: Anavex has 60 days to respond to the re-examination request, followed by 60 days of review by new rapporteurs.
"It is a 60 + 60-day period where we respond to the re-examination request, and then the review by the two rapporteurs will take another 60 days. That's why we stated that we expect this process to last for the first half of this year."
— Christopher Missling, PhD
The re-examination will include a Scientific Advisory Group (SAG) for neurology, which Anavex specifically requested.
New Data in EMA Resubmission
When asked what additional data will be included in the resubmission, management outlined a comprehensive package:
- Aβ-Clear population data – Precision medicine subset analysis
- Brain atrophy correlation – Clinical efficacy linked to reduced brain region atrophy
- COL24A1 wild-type data – Gene with estimated >70% prevalence in early AD
- Open-label extension (OLE) data – Long-term treatment outcomes
The Aβ-Clear 3 population (SIGMAR1 wild-type + COL24A1 wild-type) achieved statistical significance across all three endpoints (ADAS-Cog13, ADCS-ADL, CDR-SB) with "clinically meaningful effect sizes 2-3 times larger than other compounds on the market."
CHMP Rejection Clarification
An analyst noted apparent confusion in the CHMP rejection language regarding SIGMAR1 mutations. Management clarified the drug is more effective for SIGMAR1 wild-type patients (no mutation), contrary to how the rejection statement read. The ADCS-ADL endpoint was the only measure that didn't reach significance in the wild-type population, which management attributes to the scale not being sensitive enough for early Alzheimer's disease—a position supported by regulatory guidance.
FDA Submission Timeline
On FDA engagement, management indicated data submission is "in process" but requires coordination with formal meeting requests:
"You have to also understand the FDA has a certain meeting request which requires some time to schedule, and this is in the process as well... you have to make it in consistency with a meeting request, and that's what will happen."
— Christopher Missling, PhD
ACCESS-AD Trial Details (AD006)
Management provided specifics on the ACCESS-AD program, funded by the European Commission's Innovative Health Initiative:
Ongoing Clinical Programs
Management clarified the current clinical activity:
Currently Active:
- Compassionate use – Rett Syndrome (Canada, UK, Australia)
- Compassionate use – Alzheimer's disease
Planned Trials:
- Parkinson's disease (not yet started; prior trial was Parkinson's disease dementia)
- Fragile X Syndrome
- Schizophrenia continuation (following Phase II completion)
- One undisclosed indication
How Did the Stock React?
AVXL shares jumped 7.6% to $4.10 on the earnings release, with aftermarket activity pushing the price to $4.30. This follows a volatile period—the stock had declined from $5.02 on January 26 to $3.81 on February 5 ahead of earnings.
The stock remains well off its 52-week high of $13.99, reflecting the December EMA setback. However, today's positive reaction suggests investors are encouraged by the FDA's collaborative approach and the company's extended cash runway.
Expected Development Milestones
Management outlined an ambitious set of near-term catalysts:
Blarcamesine (ANAVEX 2-73):
- Regulatory pathway update for early Alzheimer's disease
- Parkinson's disease clinical development progress
- Regulatory and clinical trial updates for Parkinson's and Rett syndrome
- Fragile X syndrome Phase 2/3 trial design
ANAVEX 3-71:
- Advancing towards pivotal clinical studies for schizophrenia-related disorders
Scientific Presentations:
- March 10-12, 2026: Oral presentation at ICFSR26 conference at Johns Hopkins University
- Publications on precision medicine populations and biomarker correlations
Key Management Quotes
"As we have entered 2026, we continue to progress our innovative clinical pipeline with particular focus on our lead candidate, oral blarcamesine in early Alzheimer's disease. Based on our commitment to improving the lives of patients with neurological disorders, we remain excited about the therapeutic potential of oral blarcamesine."
— Christopher U. Missling, PhD, President and CEO
"We are energized by the possibility of making a meaningful impact for people living with neurological diseases, offering treatment options that are not only scalable but also far easier to administer through an oral route. By lowering barriers to access and simplifying delivery, we hope to bring innovative therapeutics to a broader population."
— Christopher U. Missling, PhD
"The Aβ-Clear 3 population... achieves significance across the board—for ADAS-Cog13, for ADCS-ADL, and for CDR-SB—and they're not only achieving significance, but they also achieve this with highly clinically meaningful effect sizes, which are sometimes 2-3 times larger than what we have seen from other compounds in the pipeline or on the market."
— Christopher U. Missling, PhD, on precision medicine data
Recent Corporate Developments
Historical EPS Performance
*Values retrieved from S&P Global
What to Watch
Near-term catalysts:
- EMA re-examination outcome – Expected first half 2026 (60+60 day process) with new rapporteur team and SAG involvement
- FDA data submission – Phase IIb/III data package in process; timing contingent on meeting scheduling
- ACCESS-AD trial (AD006) – EU-funded placebo-controlled confirmatory study
- ICFSR26 presentation – March 10-12, 2026 at Johns Hopkins University
- Publications – Aβ-Clear population, COL24A1 precision medicine, atrophy correlation data
Risks:
- Continued EMA regulatory pushback could delay European commercialization
- Clinical-stage biotech with no revenue—dependent on capital markets
- Competitive Alzheimer's landscape with Biogen's Leqembi and Eli Lilly's Kisunla already approved
This analysis was generated by Fintool AI Agent based on Anavex Life Sciences' Q1 FY2026 8-K filing, press release, and earnings call transcript dated February 9, 2026.
Related: AVXL Company Profile | Q1 FY2026 8-K Filing | Earnings Call Transcript