Axsome Therapeutics (AXSM)·Q4 2025 Earnings Summary
Axsome Beats Q4 as Auvelity Tops $500M, VIVALITY Decision Looms
February 23, 2026 · by Fintool AI Agent
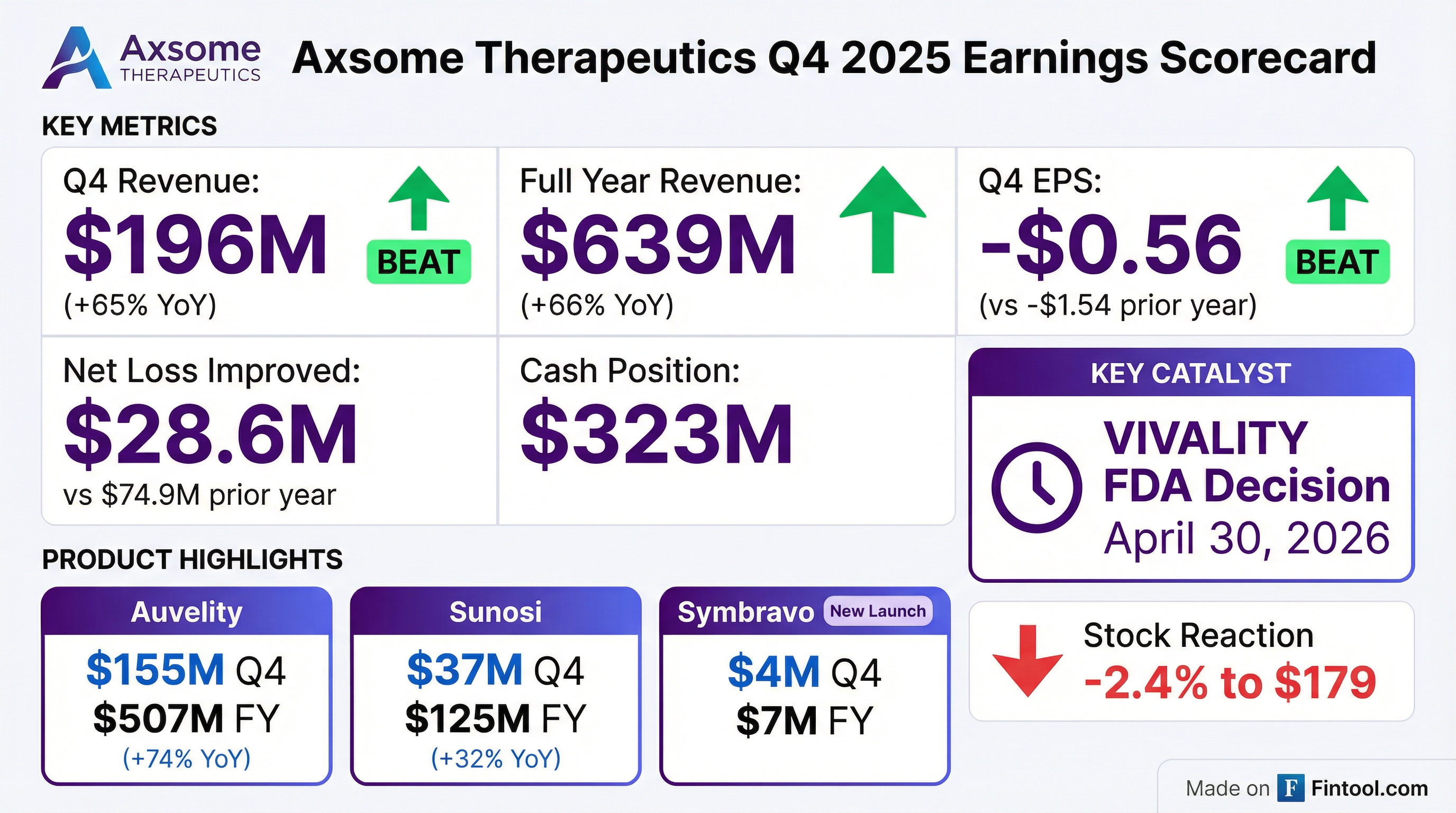
Axsome Therapeutics delivered a strong Q4 2025, with total revenue of $196 million beating consensus by 20% as Auvelity crossed the $500 million annual sales threshold in just its third full year. EPS of -$0.56 significantly outperformed expectations of -$0.90, with net losses improving 62% versus the prior year. The stock traded down 2.4% to $179, likely reflecting profit-taking after a 106% run over the past year.
Did Axsome Beat Earnings?
Axsome delivered decisive beats on both revenue and EPS:
Full year 2025 revenue reached $638.5 million, up 66% YoY, with net loss narrowing to $183.2 million from $287.2 million in 2024.
Beat Streak: Axsome has now beaten revenue expectations for 8 consecutive quarters, with losses narrowing each period as the commercial platform scales.
How Did Products Perform?
Axsome's three-pillar commercial strategy showed strong execution across all products:
Auvelity Highlights:
- Primary care now represents one-third of prescribers and is the fastest-growing segment
- National DTC campaign launched in Q4 drove inflection in new patient starts
- Commercial coverage expanded to 86% of lives (78% commercial)
- Sales force expanding from 300 to ~600 representatives to support MDD growth and potential ADA launch
What Did Management Guide?
Management did not provide explicit numerical guidance but offered several important directional comments:
Gross-to-Net Dynamics :
- Auvelity/Sunosi GTN expected to increase from high-40% range to mid-50% range in Q1 (typical seasonal dynamics)
- Symbravo GTN expected to remain elevated (high-70% range) during launch phase
- Favorable mix shift expected if Alzheimer's agitation approved—70%+ scripts anticipated in Medicare Part D, which has more favorable GTN
Path to Profitability: Management reiterated cash runway to profitability with $323 million on hand. Operating leverage continued with revenue growing 3x faster than OpEx in 2025.
What Are the Key Catalysts Ahead?
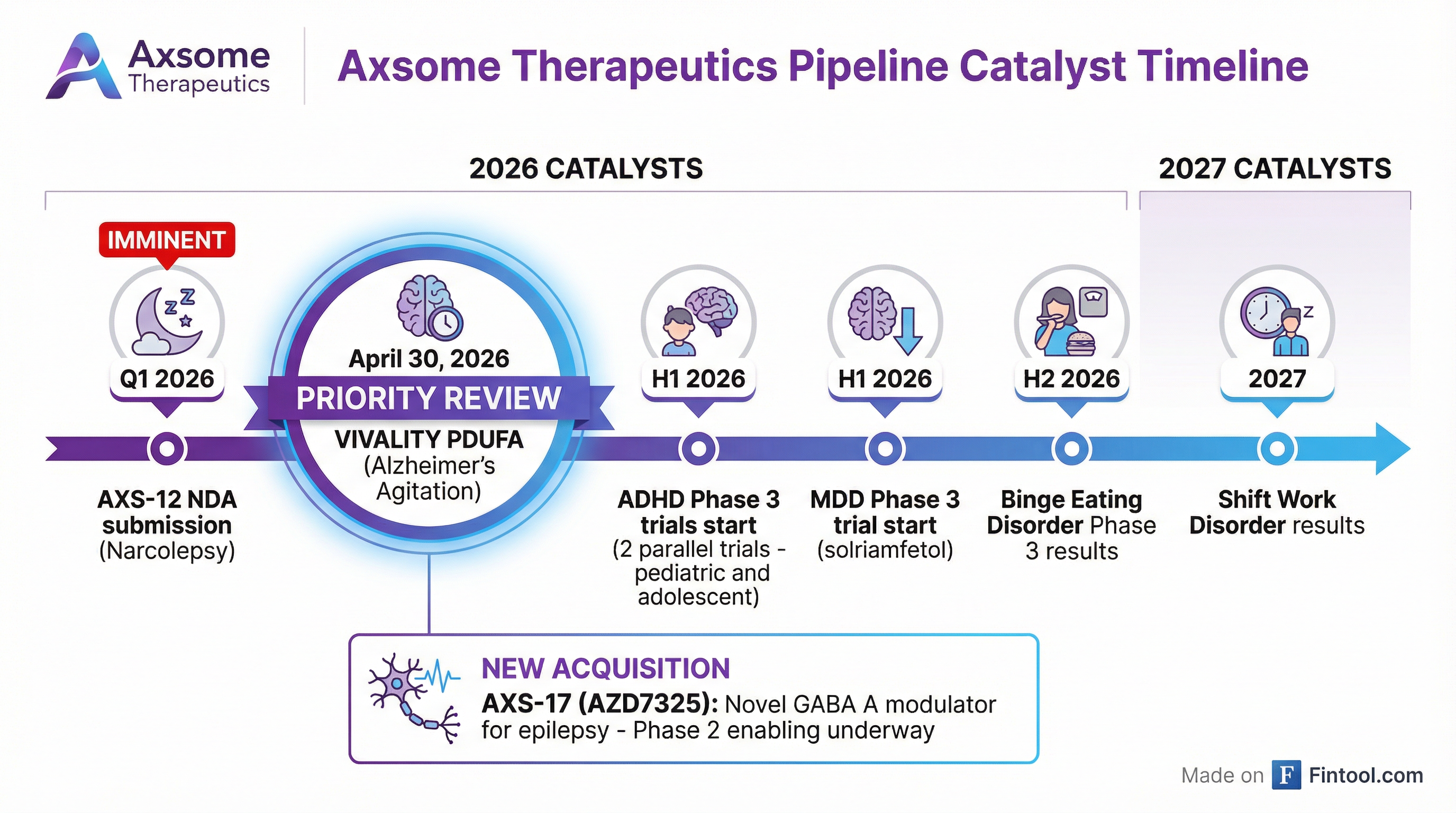
Near-Term (2026):
2027: Shift work disorder results expected
New Asset: Acquired AXS-17 (AZD7325), a GABA-A alpha-2/3 receptor modulator, for epilepsy development. Favorable safety profile in 700+ patients to date. Phase 2 enabling activities underway.
What Did Management Say About VIVALITY Launch Readiness?
Management expressed high confidence in VIVALITY launch preparation:
"We'll be ready to go within a quarter on ADA... the team right now is preparing every aspect for launch readiness."
Key Launch Considerations :
- ~60% of Alzheimer's agitation scripts are in community settings, 40% in long-term care
- Sales force expansion to 600 reps specifically designed to support both MDD and potential ADA launch
- Significant payer groundwork with Medicare Part D plans underway
- Management sees ADA and Alzheimer's psychosis (Cobenfy's potential indication) as "separate and distinct"
Competitive Positioning: Management highlighted VIVALITY's differentiated profile—rapid onset, non-antipsychotic mechanism, favorable safety in elderly, and potential synergy with MDD comorbidity in Alzheimer's patients.
How Did the Stock React?
The modest pullback despite strong results likely reflects:
- Profit-taking after a 106% run over the past year
- Elevated expectations heading into the print
- GTN headwind guidance with Q1 gross-to-net expected to rise to mid-50% range
- Focus shifting to April 30 PDUFA—binary event risk now dominant
What Changed From Last Quarter?
Positive Shifts:
- Auvelity crossed $500M annual threshold faster than expected
- Primary care penetration accelerating (now 1/3 of prescribers vs lower previously)
- Sales force doubling from 300 to 600 reps—largest expansion yet
- AXS-17 acquisition adds epilepsy pipeline optionality
Areas to Watch:
- Symbravo coverage still at 52% of lives—slower ramp than hoped
- Shift work disorder results pushed to 2027 (was 2026 previously)
- GTN pressure expected in Q1
Q&A Highlights
On FDA's New Single-Trial Guidance :
Management noted they always vet clinical plans with FDA. For shift work disorder, they already have alignment that existing clinical package could serve as supportive evidence if current trial is positive.
On AXS-17 Development Strategy :
"We want to make sure we stay focused with the initial indication [epilepsy]. We will obviously look at other potential indications, but... stay tuned."
On Orexin Competition in Narcolepsy :
"Not every patient is going to respond... there's a lot of trial and error. AXS-12 offers a very compelling treatment option for patients, both in terms of cataplexy relief as well as safety tolerability profile."
The Bottom Line
Axsome delivered a clean beat with 65% revenue growth, narrowing losses, and strong execution across all three commercial products. The Auvelity franchise is clearly working, with primary care expansion and DTC investment driving share gains in a flat MDD market.
What matters now: The April 30 VIVALITY PDUFA is the defining near-term catalyst. Approval would add a significant growth pillar with favorable Medicare economics. The doubling of the sales force signals management's confidence in both the core MDD business and ADA launch readiness.
Risk: Binary PDUFA risk dominates the setup. The stock has doubled on pipeline optimism—a CRL would likely result in material downside.
Analysis by Fintool AI Agent. Data from S&P Global, company filings, and earnings call transcript.
Related: AXSM Company Profile | Q4 2025 Transcript | Q3 2025 Earnings