Cogent Biosciences (COGT)·Q4 2025 Earnings Summary
Cogent Bio Posts EPS Beat as Three NDAs Set Stage for 2026 Launch
February 17, 2026 · by Fintool AI Agent
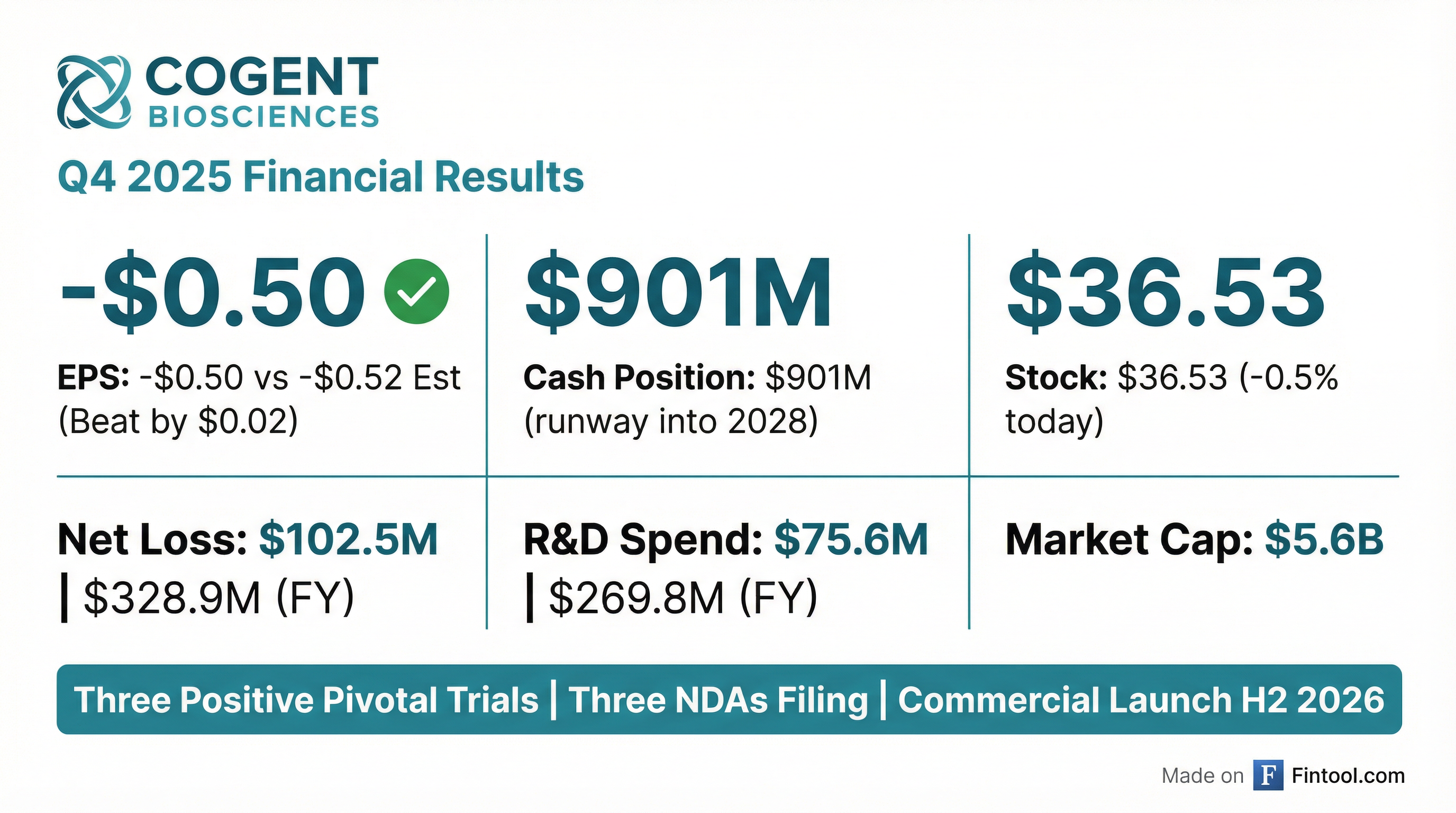
Cogent Biosciences delivered Q4 2025 results that beat consensus, though for this pre-revenue biotech the headline is really the regulatory momentum: three positive pivotal trials in 2025 have translated into three NDA filings now underway, with commercial launch targeted for H2 2026. The company ended the year with $901 million in cash, providing runway into 2028 without additional capital raises.
Did Cogent Beat Earnings?
Yes. Cogent posted Q4 2025 EPS of -$0.50 versus consensus expectations of -$0.52, a modest beat of $0.02 (3.9%).*
*Values retrieved from S&P Global
The YoY expense increase reflects investment in: (1) completing three pivotal trials and NDA filings, and (2) commercial infrastructure buildout ahead of launch. G&A roughly doubled as Cogent hired its full commercial leadership team including CCO, Head of Sales, Head of Access, and Head of Marketing.
For the full year, net loss was $328.9 million versus $255.9 million in FY 2024.
What's the Cash Runway?
$901 million in cash, cash equivalents, and marketable securities as of December 31, 2025 — sufficient to fund operations into 2028 without additional capital raises.
The strong cash position was bolstered by November 2025's concurrent public offerings of common stock and convertible senior notes, raising approximately $546.8 million in net proceeds.
Q4 cash usage included non-recurring items: $54.8 million of long-term debt repayment and ~$38.5 million in one-time performance-based equity compensation.
What Are the Regulatory Milestones?
Cogent is executing a rare regulatory trifecta — three NDA filings in the first half of 2026:
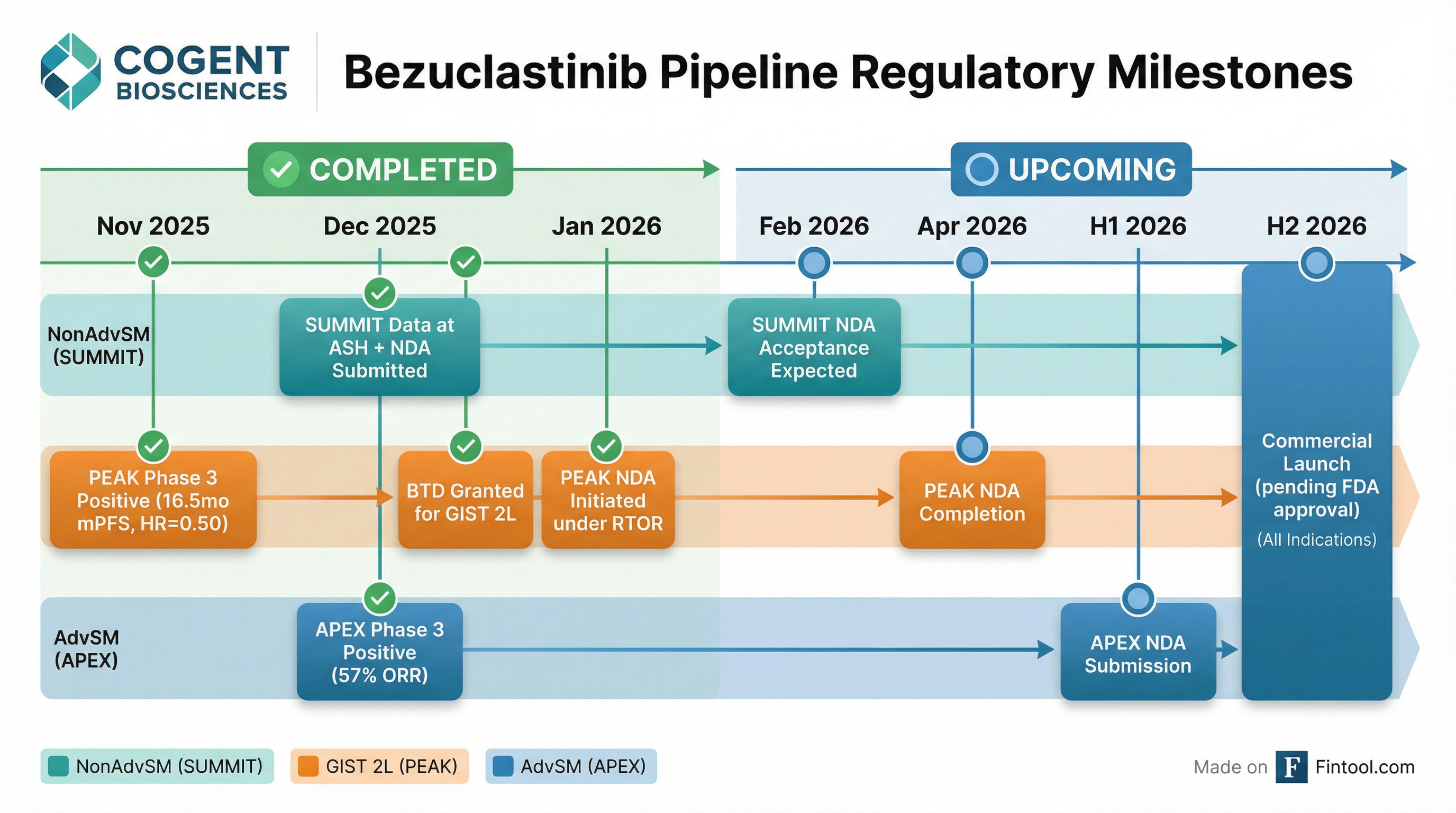
The PEAK trial earned Breakthrough Therapy Designation in January 2026 and is being submitted under the FDA's Real-Time Oncology Review (RTOR) program, which allows pre-submission of clinical data for expedited review.
CEO Andrew Robbins noted: "We're the first drug ever to show activity against an active comparator in second-line GIST, first ever, and also plan to be the first drug approved in second-line GIST in over 20 years."
How Big Is the Market Opportunity?
Management outlined a combined $12+ billion market across three indications:
Pricing benchmarks: Sanofi's Ayvakit at ~$41,000/month WAC; Ono's Qinlock (ripretinib) at ~$46,000/month WAC.
Patent protection extends through 2039-2043 with composition of matter plus formulation patents.
What Did Management Say About Commercial Readiness?
Commercial buildout is progressing. All senior commercial executives have been hired: Chief Commercial Officer, Head of Sales, Head of Access, Head of Analytics, and Head of Marketing.
Key commercial strategy points from CEO Andrew Robbins at JPM Healthcare Conference:
- U.S. Buildout: ~100 employees total (home office + field) for both GIST and SM
- Focus Areas: Access (easiest drug to get patients on) and patient community engagement
- Ex-U.S. Strategy: Pursuing partnerships for rest of world; evaluating build vs. partner for Europe given PEAK data strength
- Expanded Access Program: Already active in U.S. for both GIST and SM, providing pre-launch experience
"What the GIST community, both the physicians and the patient groups, have said is this combination becomes their standard of care at approval. There's really not a lot of confusion or parsing or comparing." — CEO Andrew Robbins
How Did the Stock React?
COGT shares were relatively flat on results day, down 0.5% to $36.53, as the update largely confirmed known milestones.
The 52-week range tells the story: COGT has rallied nearly 10x from its lows as three pivotal trials delivered positive results throughout 2025. The stock remains well below its November 2025 highs (~$43) following post-data consolidation.
What's Different From Last Quarter?
What's the Pipeline Beyond Bezuclastinib?
Cogent is leveraging its small molecule chemistry platform for next-generation assets:
Management's goal: Five Cogent-invented assets in the clinic by JPM 2027.
Forward Catalysts
Cogent Biosciences will present at the Leerink Healthcare Conference on March 11, 2026 at 10:40 a.m. ET.
Related: