Jazz Pharmaceuticals (JAZZ)·Q4 2025 Earnings Summary
Jazz Pharma Beats Q4 on Record Revenue, Raises 2026 Outlook
February 24, 2026 · by Fintool AI Agent
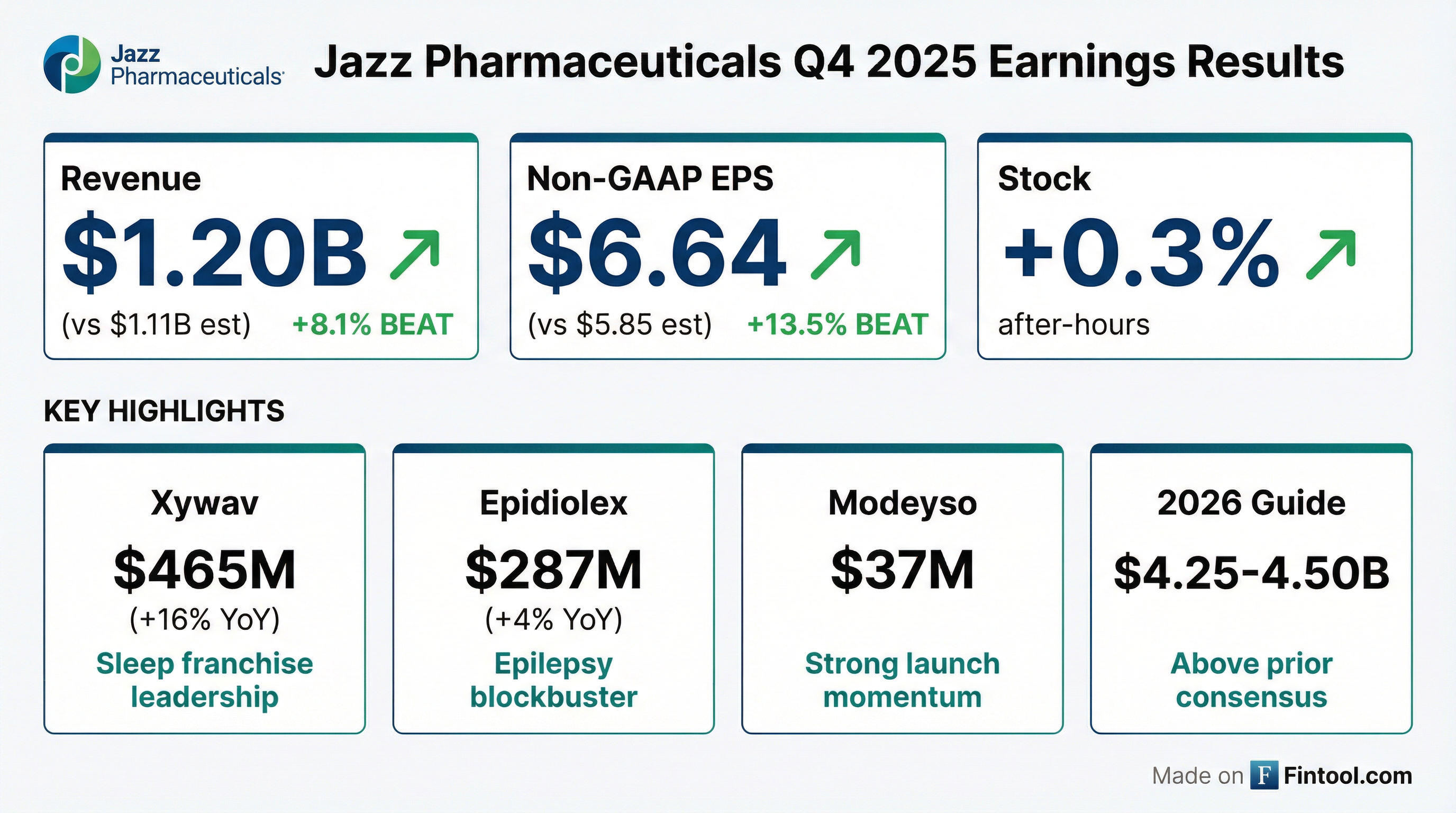
Jazz Pharmaceuticals delivered its highest revenue quarter ever with Q4 2025 results that beat both top and bottom line estimates. Total revenues of $1.20 billion grew 10% year-over-year, driven by continued strength in the sleep franchise and a strong Modeyso launch . The company guided 2026 revenues of $4.25-4.50 billion, reflecting confidence in its core franchises and upcoming zanidatamab launch .
Did Jazz Pharmaceuticals Beat Earnings?
Jazz delivered a solid beat on both metrics:
Full-year 2025 revenue reached a record $4.27 billion, up 5% year-over-year, marking Jazz's 21st consecutive year of top-line growth . The company generated $1.4 billion in cash from operations, demonstrating strong conversion of revenue to cash flow .
How Did the Stock React?
Despite the strong beat, JAZZ stock was relatively muted, rising just +0.3% in after-hours trading to $173.79. The stock has rallied significantly from its 52-week low of $95.49, now trading near its 52-week high of $182.99.
The muted reaction may reflect that much of the good news was already priced in — JAZZ shares have gained ~82% from their 52-week low, outperforming the broader healthcare sector.
What Did Management Guide?
Jazz provided 2026 guidance that implies continued growth with a significant pipeline catalyst ahead:
Key guidance drivers :
- Xywav: Flat to up mid-single digits (continued patient growth)
- Epilepsy + Oncology: Double-digit combined growth
- Modeyso: First full year of commercial sales
- Ziihera (zanidatamab): Potential 2H26 launch in first-line HER2+ GEA

What Changed From Last Quarter?
Several notable developments this quarter:
Positive Changes:
- Record revenue quarter — $1.20B was Jazz's highest quarterly revenue ever
- Modeyso launch momentum — $37M in Q4 (first full quarter), $48M since August 2025 launch
- Zanidatamab FDA Breakthrough Therapy Designation for HER2+ GEA
- R&D expense decline — Down due to zanidatamab trial timing and JZP385 discontinuation
Headwinds:
- Litigation settlements — $234M Xyrem antitrust settlement and $90M Avadel settlement impacted SG&A
- Xyrem decline — $38M in Q4 vs $49M prior year as generic competition continues
- Vyxeos weakness — Down to $35M from $53M in Q4 2024
Product Performance Deep Dive
Neuroscience Franchise ($848M Q4, +8% YoY)
Xywav highlights :
- ~500 net patient adds in Q4
- 16,175 active patients (10,950 narcolepsy + 5,225 IH)
- Sleep franchise exceeded $2 billion in 2025 (including AG royalties)
Epidiolex achieved blockbuster status in 2025 with over $1.1 billion in annual revenue, up 9% YoY .
Oncology Franchise ($337M Q4, +16% YoY)
Modeyso (dordaviprone) was acquired through the Chimerix acquisition and launched in August 2025 for H3 K27M-mutant diffuse midline glioma. Jazz sold the associated Rare Pediatric Disease Priority Review Voucher for $200M gross proceeds (50% to Jazz) .
Pipeline Catalysts to Watch
Zanidatamab (Ziihera) — Jazz's most important near-term catalyst:
CEO Renee Gala highlighted that the Phase 3 HERIZON-GEA-01 results "firmly position zanidatamab as the HER2-targeted agent of choice" regardless of PD-L1 status, which "de-risks our clinical trials in additional indications, including HER2+ metastatic breast cancer" .
Balance Sheet & Capital Allocation
Notable: Jazz appointed Tom Riga as Chief Business Officer to "accelerate corporate development efforts across rare disease" , signaling continued M&A appetite despite elevated leverage.
Key Risks Flagged
Management highlighted several forward-looking risks :
- Regulatory risk — sBLA for zanidatamab may not be approved on expected timeline
- Generic competition — Xyrem continues to decline; potential generics to Epidiolex
- Reimbursement — Obtaining coverage for specialty products remains challenging
- Pipeline execution — Clinical trials may fail to confirm benefit or enable regulatory submissions
- Integration risk — Chimerix integration still ongoing
The Bottom Line
Jazz delivered a clean beat in Q4 2025 with record quarterly revenue and strong execution across key franchises. The 2026 guidance implies modest top-line growth with significant optionality from zanidatamab, which could launch in HER2+ gastric cancer in 2H26. While the stock reaction was muted (shares have already rallied 82% from lows), the fundamental story remains intact: diversified rare disease portfolio generating strong cash flows with a de-risked pipeline catalyst ahead.
What to watch next:
- Q1 2026: Zanidatamab sBLA submission confirmation
- 2H 2026: Zanidatamab GEA launch timing
- Modeyso trajectory in first full year
Data sourced from Jazz Pharmaceuticals Q4 2025 8-K filing and S&P Global consensus estimates.