Tarsus Pharmaceuticals (TARS)·Q4 2025 Earnings Summary
Tarsus Q4 2025: Revenue Beats, 2026 Guidance of $670-700M Sends Shares Soaring 9%
February 23, 2026 · by Fintool AI Agent

Tarsus Pharmaceuticals delivered blockbuster Q4 2025 revenue of $151.7 million, beating estimates by 5.5%, and shares surged ~9% in aftermarket trading to $76.04 after management provided 2026 guidance for the first time: $670-700 million in net product sales, representing 50%+ growth at the midpoint . Full-year 2025 XDEMVY sales reached $451.4 million at a 45% gross-to-net discount, with peak U.S. sales potential now exceeding $2 billion .
Did Tarsus Beat Earnings?
Revenue: Beat by 5.5% | EPS: Missed by 185.7%
The revenue beat reflects XDEMVY's exceptional commercial momentum, with 130,000 bottles delivered to patients in Q4 versus 58,500 in Q4 2024—a 122% year-over-year increase . However, the EPS miss stems from a significant ramp in selling, general & administrative expenses, which ballooned to $130.7 million in Q4 2025 from $69.0 million a year ago .
What Drove the EPS Miss?
The $61.7 million year-over-year increase in SG&A was driven by three main factors :
Stock-based compensation also increased materially, with SG&A non-cash comp reaching $10.5 million versus $5.5 million in Q4 2024 .
How Did the Stock React?
TARS shares closed at $69.59 on February 23, 2026, up 4.5% during regular trading. Aftermarket trading surged 9.3% to $76.04 as investors cheered the first-ever full-year guidance and raised peak sales potential.
52-Week Range: $38.51 - $85.25 | Market Cap: ~$3.0B
The positive aftermarket reaction contrasts with the 4.6% decline after Q3 2025 results, suggesting the 2026 guidance of $670-700M (+50% YoY) and $2B+ peak sales commentary provided the clarity investors were looking for on XDEMVY's long-term trajectory.
What Changed From Last Quarter?
Key sequential improvements:
- Revenue acceleration: Q4 marked the 8th consecutive quarter of growth, with revenue up 28% sequentially versus 16% QoQ in Q3
- Loss narrowing: Net loss improved from $12.6M to $8.4M despite higher spend, showing operating leverage
- DTC momentum: Unaided awareness of Demodex blepharitis reached 25% versus just 2% when the campaign began
- Prescriber depth: 40% of core target now prescribing weekly; daily writers grew 20% QoQ
- Retreatment trends: Weekly refills trending in low-to-mid teens, moving toward 20% steady-state target
XDEMVY Growth Trajectory
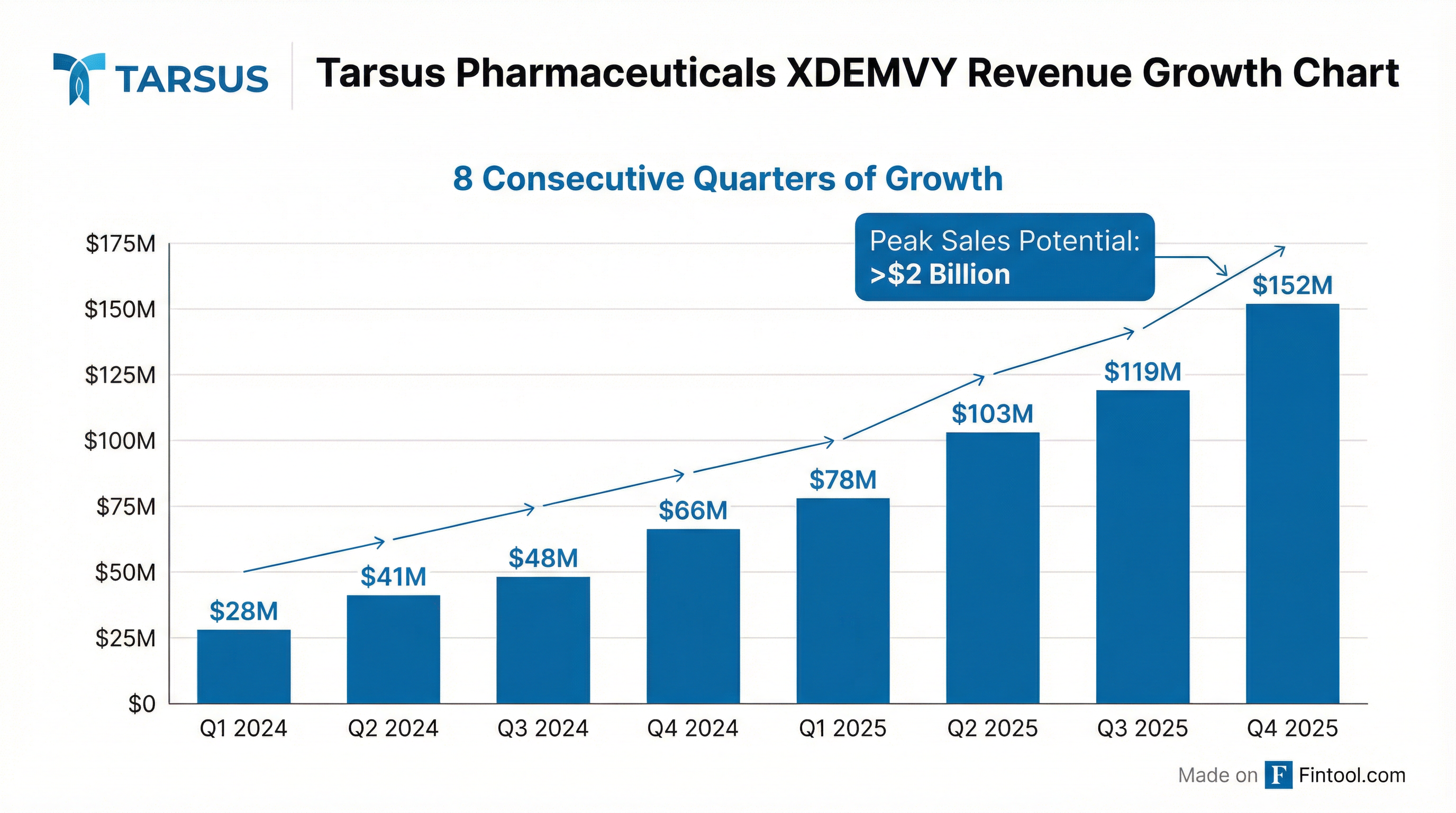
XDEMVY has delivered 8 consecutive quarters of sequential revenue growth:
Full-Year 2025: $451.4 million in net product sales (+151% YoY), with approximately 400,000 bottles delivered to patients .
What Did Management Guide?
For the first time, Tarsus provided full-year 2026 guidance:
Quarterly Cadence: Management warned that Q1 2026 revenue will be flat to slightly below Q4 2025 due to deductible resets, weather disruptions, and typical seasonality . Growth is expected to follow 2025 patterns: strong Q2, tempered Q3, robust Q4.
Peak Sales Potential: Management raised XDEMVY's U.S. peak sales potential to exceed $2 billion, citing :
- 500,000+ patients treated since launch—less than 10% of the 25M Americans with DB
- 90%+ payer coverage across commercial, Medicare, and Medicaid
- Fundamental shift in how physicians diagnose and treat Demodex blepharitis
- DTC campaign driving 25% unaided awareness (up from 2% at campaign start)
CEO Bobak Azamian stated: "In just two years since launch, XDEMVY has driven a fundamental shift in eye care... We see a clear path to peak sales potential of more than $2 billion."
Global Expansion Timeline
CFO Jeff Farrow noted ex-U.S. opportunity could add ~10% to U.S. peak sales, with Japan potentially larger than Europe .
Q&A Highlights
On Peak Sales Timing: CEO Azamian indicated the company is "about a couple of years from $1 billion-plus" with "no signs of slowing down," though declined to pinpoint exact timing for $2B peak .
On Seasonality: CCO Mottiwala confirmed XDEMVY is now subject to "typical seasonality" seen across eye care, with Q1 impacted by deductible resets affecting both patient visits and gross-to-net. IQVIA data in recent weeks shows "positive trajectory" already recovering .
On DTC Spend: Management will maintain similar ~$80M DTC spend in 2026, noting ROI is positive and improving due to compounding effects of 25% awareness plus physician pull-through .
On OpEx Step-Ups: CFO Farrow noted "no material step-ups in the out years" beyond variable costs that scale with revenue (pharmacy fees, co-pay programs). DTC spend could potentially be "pared back or pulsed" in 2027+ .
On Ex-U.S. Opportunity: A "good proxy" is 90% U.S. / 10% rest of world, with Japan potentially larger than Europe given pricing/reimbursement dynamics .
Pipeline Catalysts Ahead
Tarsus is applying its "category creation" playbook to two additional high-unmet-need diseases:
Ocular Rosacea affects approximately 15-18 million Americans with no FDA-approved treatments. TP-04 is a novel lotilaner-based ophthalmic gel targeting erythema and telangiectasias around the eye .
TP-05 is a ~700-patient Phase 2 trial enrolling in one tick season. The oral tablet is designed to kill Lyme-infected ticks within 24 hours before disease transmission—95%+ tick killing activity demonstrated in prior studies .
Balance Sheet & Cash Position
The substantial cash position (~$418M) provides runway to fund continued commercial investment, pipeline advancement (TP-04 and TP-05 Phase 2 trials), and global expansion without near-term dilution risk.
2026 Investment Priorities
- Sales Force Expansion: Adding 15-20 key account leaders targeting high-opportunity practices; expected to contribute meaningfully in 2H 2026
- DTC Campaign: ~$80M spend maintained, with focus on highest-ROI channels and formats
- Pipeline R&D: $115-135M including TP-04 ($7-10M) and TP-05 ($25-30M) Phase 2 trials
Board Addition: David Pyott
Tarsus announced that David Pyott, former Chairman and CEO of Allergan, joined the Board of Directors . Pyott led Allergan for 17 years and built global eye care franchises including Restasis and Botox. CEO Azamian noted his experience "building enduring global eye care franchises and driving disciplined growth at scale will be invaluable as we continue to expand Tarsus' reach."
Key Risks and Concerns
-
Near-Term Profitability: XDEMVY is now profitable at the product line level, but company-level profitability remains ahead. Management maintains "clear line of sight" but hasn't provided a specific timeline .
-
Q1 Seasonality: Guidance for Q1 2026 to be flat-to-down versus Q4 2025 may disappoint investors looking for linear growth .
-
DTC Spending Dependency: The company is investing ~$80M annually in DTC advertising . While generating positive ROI, any pullback could impact growth trajectory.
-
Pipeline Execution: TP-04 and TP-05 Phase 2 trials represent $32-40M in R&D spend with results in 1H 2027. Failure could limit the company's long-term growth story beyond XDEMVY.
-
Competition Risk: While XDEMVY is currently the only FDA-approved Demodex treatment, success could attract competition in this newly validated market.
Historical Earnings Performance
Tarsus has beaten revenue estimates in all 5 quarters shown, but EPS results have been mixed, with 2 misses in the past 3 quarters. The pattern suggests management is prioritizing growth investment over near-term profitability.
The Bottom Line
Tarsus delivered another quarter of exceptional revenue growth (+128% YoY) and, critically, provided first-ever full-year guidance of $670-700M for 2026—representing 50%+ growth and validating the XDEMVY commercial trajectory. Management's raised U.S. peak sales target to $2B+ signals confidence that the product has blockbuster-plus potential with only ~10% of the 25M addressable patients treated so far.
The 9% aftermarket rally suggests investors welcomed the guidance clarity and long-term visibility that was missing in prior quarters. While Q1 will be seasonally soft, the fundamental story remains intact: strong prescriber momentum (40% writing weekly), growing patient awareness (25% unaided), and a pipeline with two Phase 2 trials reading out in 1H 2027.
Key 2026 Catalysts:
- Q2: TP-05 (Lyme disease) Phase 2 initiation
- H2: 15-20 new key account leaders driving depth
- 2H 2026: China approval via Grand Pharmaceutical
- 1H 2027: TP-04 and TP-05 Phase 2 topline data
Data sources: Tarsus Pharmaceuticals Q4 2025 earnings call transcript, 8-K filing, S&P Global estimates. Stock prices as of February 23, 2026.