Blackstone and J&J Strike First-Ever Co-Funding Deal for AML Drug
February 23, 2026 · by Fintool Agent

Blackstone Life Sciences and Johnson & Johnson have entered their first-ever co-funding agreement, partnering to finance clinical development of bleximenib, an investigational oral menin inhibitor for acute myeloid leukemia (AML). The deal marks a novel financing structure in biopharma and positions bleximenib to compete in an increasingly crowded menin inhibitor market where two rivals have already secured FDA approval.
The partnership reflects both companies' strategic priorities: Blackstone's expansion of its $15 billion life sciences platform through corporate partnerships, and J&J's drive to become the number one oncology company with a $50 billion revenue target by decade's end.
Deal Structure: Risk-Sharing in Drug Development
Under the agreement, J&J and funds managed by Blackstone Life Sciences will jointly finance a portion of ongoing and future clinical trials of bleximenib in AML. While financial terms were not disclosed, the structure represents Blackstone's "corporate partnership" model—risk-sharing collaborations with biopharmaceutical partners on drug development programs.
"We believe that bleximenib's promising clinical data, combined with Johnson & Johnson's deep expertise in hematologic malignancies, create a strong foundation to address critical gaps in patient care," said Dr. Nicholas Galakatos, Global Head of Blackstone Life Sciences.
Dr. Ari Brettman, Senior Managing Director at BXLS, emphasized the urgency: "As an aggressive, fast-progressing blood cancer with high relapse rates, there is an urgent need for better, more tolerable treatment options for patients living with AML."
The AML Opportunity: Aggressive Cancer, Unmet Need
AML is the most common type of acute leukemia in adults, affecting approximately 20 million people globally annually—including 2 million Americans—with that number expected to rise. The disease is extremely challenging to treat and carries the lowest survival rate of all leukemias. Without prompt treatment, patients can die within months.
Bleximenib targets the menin-KMT2A protein interaction, disrupting a key oncogenic pathway that drives leukemic cell growth in patients with KMT2A gene rearrangements or NPM1 mutations—genetic variants found in approximately 35-40% of AML cases combined.
The drug is currently being investigated in Phase 1, 2, and 3 clinical trials, both as monotherapy and in combination with other AML-directed therapies. Early clinical data has shown encouraging response rates:
| Metric | Bleximenib (Early Data) |
|---|---|
| Overall Response Rate | 48-50% |
| CR/CRh Rate | 33% |
| Differentiation Syndrome | 13% (Grade ≥3: 7%) |
| QTc Prolongation | 1% (Grade ≥3: 1%) |
Notably, bleximenib appears to have a favorable safety profile on cardiac side effects compared to competitors, with minimal QTc prolongation—a key differentiator as clinicians weigh treatment options for often elderly, comorbid patients.
Entering a Competitive Menin Race
Bleximenib enters the market behind two FDA-approved menin inhibitors, creating both competitive pressure and validation for the mechanism:
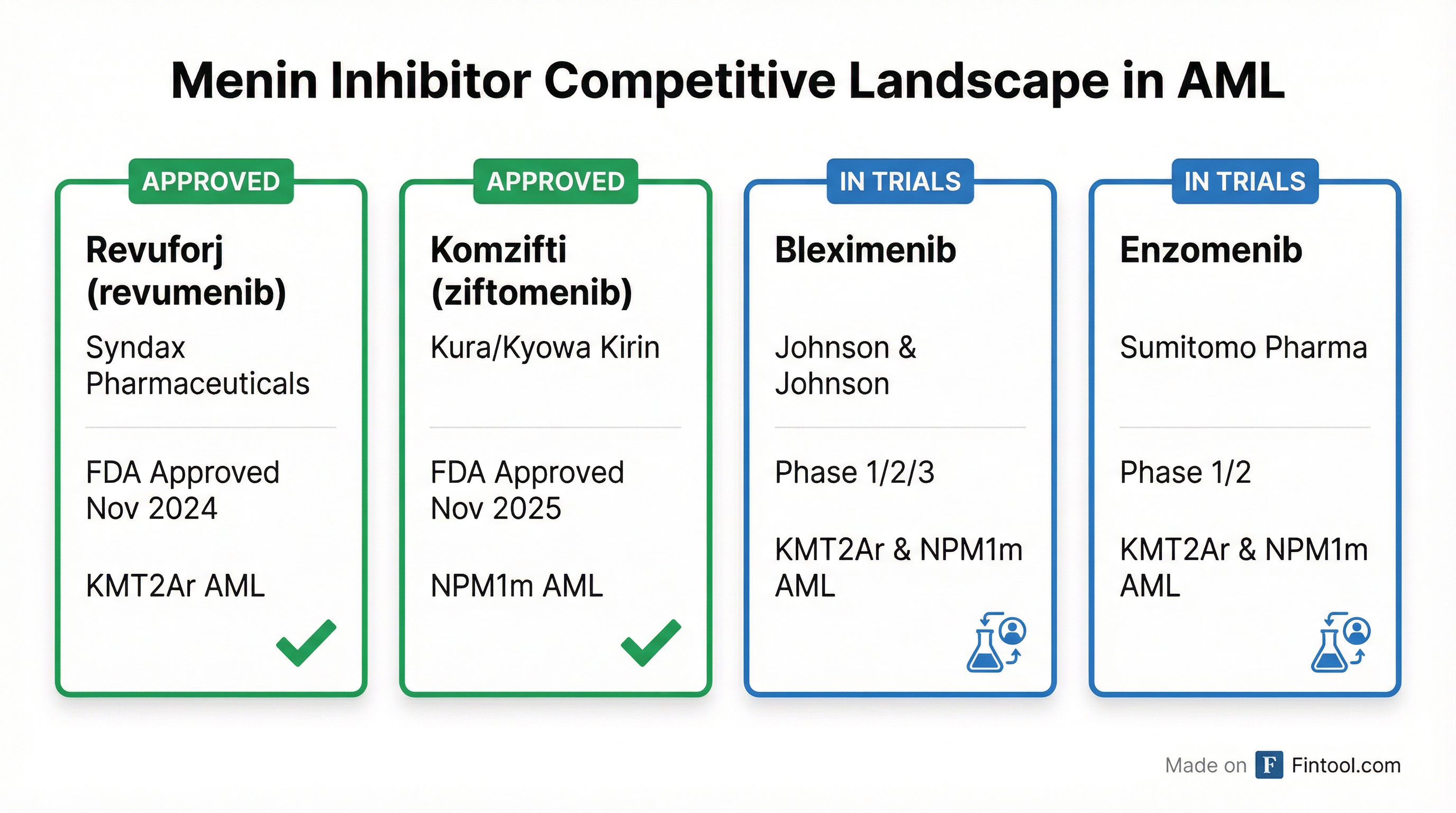
Revuforj (revumenib) from Syndax Pharmaceuticals received FDA approval in November 2024 for relapsed/refractory acute leukemia with KMT2A translocation, followed by an expanded approval for NPM1-mutant AML in October 2025. The drug carries boxed warnings for both differentiation syndrome and QTc prolongation.
Komzifti (ziftomenib) from Kura Oncology and Kyowa Kirin was approved in November 2025 for relapsed/refractory NPM1-mutant AML. It avoided the boxed warning for QTc prolongation and offers once-daily dosing versus Revuforj's twice-daily regimen.
The next battleground is first-line AML, where combination studies with standard-of-care chemotherapy are underway. J&J's deep expertise in hematology—particularly its dominant position in multiple myeloma with Darzalex, Carvykti, Tecvayli, and Talvey—provides commercial infrastructure advantages as bleximenib advances.
Strategic Rationale: Why This Deal, Why Now
For Blackstone
Blackstone Life Sciences manages $15 billion in assets under management across its life sciences platform, which invests across the life cycle of companies and products in the pharmaceutical, biotechnology, and medical technology sectors.
The firm held initial closings of $1.6 billion for its new Life Sciences flagship fund in late 2024, targeting at least the size of its prior $5 billion fund. Corporate partnership transactions like the J&J deal offer:
- Risk-sharing: Co-funding reduces capital at risk while maintaining upside through milestone-based payments
- Strategic access: Partnerships with large pharma provide deal flow and validation for future investments
- Diversification: The life sciences portfolio complements Blackstone's real estate, credit, and private equity businesses
| Blackstone Financials | Q4 2025 | Q3 2025 | Q2 2025 | Q1 2025 |
|---|---|---|---|---|
| Revenue ($B) | $2.7* | $2.2 | $2.7* | $2.4* |
| Net Income ($B) | $1.0 | $0.6 | $0.8 | $0.6 |
| Total Assets ($B) | $47.7 | $46.6 | $45.4 | $45.3 |
*Values retrieved from S&P Global
For Johnson & Johnson
J&J is executing an aggressive transformation toward higher-growth segments, with oncology targeted to reach $50 billion in sales by decade's end. The company's Q4 2025 earnings call emphasized six areas of strategic focus, with oncology first on the list.
The partnership allows J&J to:
- Share development costs: Late-stage clinical trials are expensive; co-funding reduces capital requirements
- Validate pipeline assets: External investment signals confidence in bleximenib's potential
- Maintain optionality: J&J retains full commercial rights while reducing R&D burn
| J&J Financials | Q4 2025 | Q3 2025 | Q2 2025 | Q1 2025 |
|---|---|---|---|---|
| Revenue ($B) | $24.6 | $24.0 | $23.7 | $21.9 |
| Net Income ($B) | $5.1 | $5.2 | $5.5 | $11.0 |
| Cash ($B) | $19.7 | $18.2 | $18.6 | $38.5 |
CEO Joaquin Duato laid out the oncology ambition clearly: "Our ambition with oncology is to become the number one oncology company, reaching $50 billion by the end of the decade, sustained by our success in multiple myeloma and also in solid tumors with lung cancer, prostate cancer, and bladder cancer."
What to Watch
Clinical milestones: Bleximenib Phase 3 data readouts will determine whether the drug can differentiate on efficacy or safety versus approved competitors. The combination data with azacitidine/venetoclax in newly diagnosed AML is particularly important for first-line market positioning.
Regulatory pathway: With two competitors already approved, bleximenib's path may be clearer but also more competitive. Any safety advantages (particularly on QTc prolongation) could drive prescribing preferences.
Commercial execution: J&J's existing oncology infrastructure and hematology sales force represent a meaningful competitive advantage over smaller biotech competitors.
Deal economics: While terms weren't disclosed, investors should watch for any SEC filings that reveal the magnitude of Blackstone's commitment and milestone-based payment structures.
The Bottom Line
The Blackstone-J&J deal represents a novel financing model that may become more common as drug development costs rise and pharma companies seek to share risk on pipeline assets. For Blackstone, it's another step in building out its life sciences platform alongside traditional private equity. For J&J, it validates bleximenib's potential while freeing capital for other pipeline investments.
The real test comes in the clinic. With two menin inhibitors already approved, bleximenib needs to demonstrate either superior efficacy, a better safety profile, or compelling combination data to carve out meaningful market share. The early data on cardiac safety is encouraging, but the Phase 3 readouts will be definitive.
For investors, this deal is worth monitoring as a signal of evolving pharma-private equity relationships and as a window into the competitive dynamics of the AML market—one of the most challenging and fast-moving areas of oncology.
Related Companies: Blackstone Inc. | Johnson & Johnson