GRAIL Stock Crashes 46% as Landmark Cancer Screening Trial Misses Primary Endpoint
February 20, 2026 · by Fintool Agent
Grail shares collapsed 46% to around $55 Friday after the company disclosed that its landmark NHS-Galleri trial—the largest-ever randomized controlled study of a multi-cancer early detection test—failed to meet its primary endpoint of a statistically significant reduction in Stage III and Stage IV cancer diagnoses .
The trial result triggered an instant $1 billion market cap wipeout, with GRAL falling from $101.53 at Thursday's close to as low as $48 in early trading. The stock had traded above $118 just weeks ago on optimism surrounding the company's FDA submission.
The Trial Miss
The NHS-Galleri trial enrolled 142,000 participants aged 50-77 across England over three years—the most ambitious study ever conducted for a multi-cancer early detection (MCED) blood test . The primary endpoint targeted a statistically significant reduction in combined Stage III and Stage IV cancer diagnoses compared to standard NHS screening.
That endpoint was not achieved .
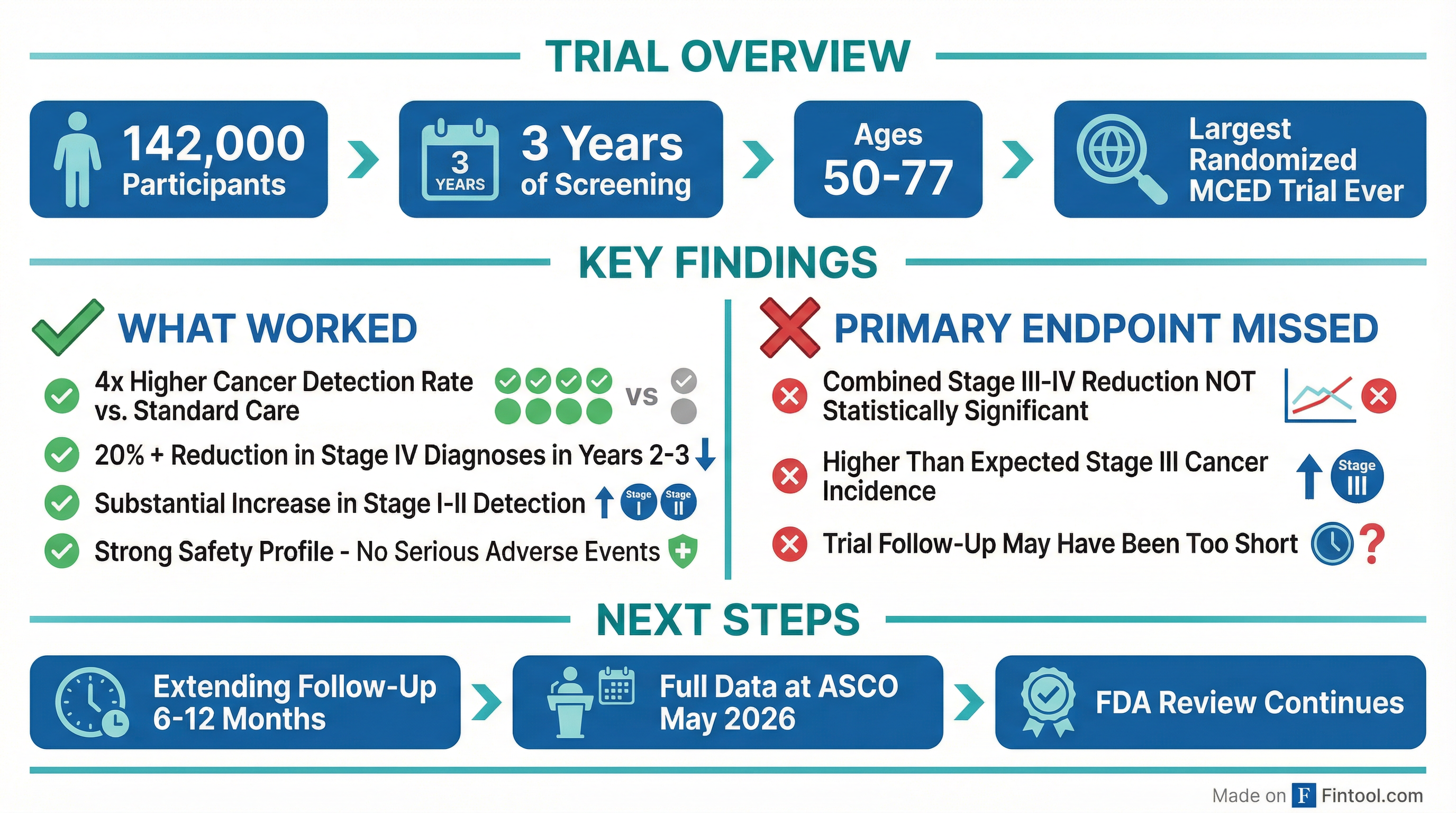
However, the data told a more nuanced story. Management highlighted several positive findings:
| Metric | Result | Context |
|---|---|---|
| Stage IV Reduction (12 Deadly Cancers) | >20% in Years 2-3 | Clinically meaningful for survival |
| Cancer Detection Rate vs Standard Care | 4x improvement | Strong detection performance |
| Stage I-II Detection | Substantial increase | Earlier detection of typically late-stage cancers |
| Safety Events | None serious | 70,000 participants tested |
"The reduction in Stage IV cancer diagnoses is a critically important outcome," said Sir Harpal Kumar, GRAIL's Chief Scientific Officer. "When cancer is detected before distant metastatic spread, we can often treat with curative intent" .
Why the Primary Endpoint Missed
The trial saw higher-than-anticipated Stage III cancer incidence, which offset the Stage IV reductions when combined . Management suggested the three-year follow-up period may have been insufficient for the control arm to fully manifest cancers that would have been caught earlier in the screening arm .
"What you tend to see in a screening trial is that you're pulling forward into your intervention arm cancers from the future," Kumar explained on the earnings call. "For a control arm of a study, those cancers may not yet have manifested" .
GRAIL plans to extend the trial's follow-up period by 6-12 months to capture more mature data. Full results will be presented at ASCO in late May .
FDA Approval: Management Says Probability Unchanged
The central question for investors: Does this miss derail FDA approval?
GRAIL executives pushed back firmly. "Everything we've learned from the FDA, their history with us, our conversations, has been their focus is gonna be on clinical performance and safety," said Josh Ofman, GRAIL's President .
The company's Premarket Approval (PMA) application, submitted in January, includes:
- First 25,000 participants from the U.S.-based PATHFINDER 2 study
- First-year performance data from NHS-Galleri (prevalence screening round)
- Bridging analysis comparing trial version to commercial version
Critically, the FDA submission focuses on clinical validation metrics—not the clinical utility endpoint that missed. The agency designated Galleri as a Breakthrough Device in 2018, and management expects approximately 12 months for review .
"Based on their prior advisory committee that they've already held, and the fact that we've addressed all of those issues in our submission... there's likely no need for" another AdCom, Ofman said .
Financial Position Remains Strong
| Metric | Q4 2025 | FY 2025 |
|---|---|---|
| Total Revenue | $43.6M | $147.2M |
| U.S. Galleri Revenue | $41.3M | $136.8M |
| Revenue Growth (YoY) | +14% | +17% |
| Net Loss | $(99.2M) | $(408.4M) |
| Cash Position | $904.4M | — |
GRAIL sold more than 185,000 Galleri tests in 2025, growing its prescriber base 30% to approximately 17,000 providers . The company raised $436 million through a private placement and ATM program in late 2025, extending its cash runway into 2030 .
Management reiterated 2026 guidance of 22-32% Galleri sales growth and cash burn of no more than $300 million .
Notably, despite the trial miss, GRAIL announced it is expanding its U.S. sales force based on the "compelling" Stage IV reduction and cancer detection rate improvements .
What to Watch
Near-Term Catalysts:
- FDA review decision (expected ~early 2027)
- Extended NHS-Galleri follow-up data (additional 6-12 months)
- Full trial results presentation at ASCO (May 2026)
Key Questions:
- Will FDA approval odds truly hold steady given the high-profile miss?
- Can the extended follow-up salvage the Stage III-IV endpoint?
- How will NHS England respond regarding a potential national screening program?
The Medicare coverage pathway established by the Nancy Gardner Sewell Medicare Multi-Cancer Early Detection Screening Coverage Act, signed into law earlier this year, remains intact for FDA-approved tests . GRAIL's REACH study in Medicare patients could provide additional clinical utility evidence for CMS consideration .
Competitive Landscape
GRAIL's trial miss comes as the MCED space heats up. Exact Sciences ($19.7B market cap) and Guardant Health ($13.8B) are developing competing liquid biopsy platforms, while Tempus Ai ($10.0B) offers AI-driven diagnostic services.
Galleri remains the only MCED test to have completed a randomized controlled trial of this scale—a potential differentiator even with the endpoint miss. The test's ability to detect 50+ cancer types with a single blood draw and provide Cancer Signal Origin localization remains unique in the market .
Related: Grail Company Profile | Exact Sciences | Guardant Health | Tempus Ai