Johnson & Johnson Bets $1 Billion on Pennsylvania Cell Therapy Plant as Carvykti Sales Near $2 Billion
February 18, 2026 · by Fintool Agent

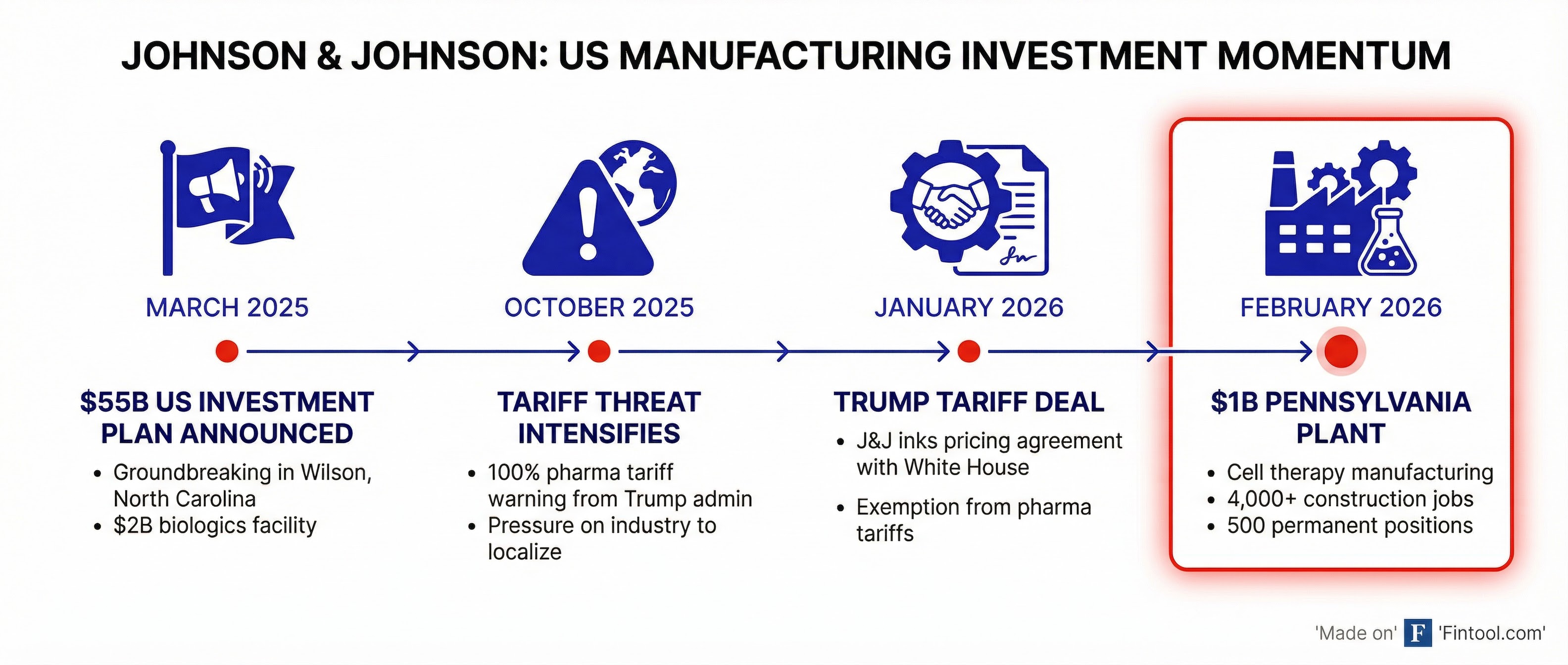
Johnson & Johnson is investing more than $1 billion to build a next-generation cell therapy manufacturing facility in Pennsylvania, the latest milestone in a sweeping domestic production buildout that has secured the pharma giant an exemption from President Trump's pharmaceutical tariffs.
The Montgomery County facility — to be operated by J&J's Janssen Biotech subsidiary at its campus near Ambler — will create more than 4,000 construction jobs and 500 permanent biomanufacturing positions once operational in 2031. Pennsylvania is contributing $41.5 million in incentives to support the project.
"Our region is home to world-class healthcare, science and research — and the opening of Johnson & Johnson's cell therapy manufacturing facility in Lower Gwynedd expands its long tradition of leadership in Pennsylvania," said Representative Madeleine Dean.
Shares of J&J rose 0.6% Wednesday to $244.83, near their 52-week high of $246.35.
$55 Billion Investment Secures Tariff Exemption
The Pennsylvania plant announcement extends J&J's $55 billion U.S. investment commitment through early 2029 — a 25% increase over the prior four years — which the company unveiled in March 2025 amid mounting tariff pressure from the Trump administration.
That gamble paid off in January 2026, when J&J negotiated a voluntary agreement with the White House that exempted its pharmaceutical products from the 100% import tariffs President Trump imposed on branded, patented drugs in September 2025. The deal came with commitments to lower prices for millions of Americans and manufacture "the vast majority" of its advanced medicines domestically.
The investment blueprint now includes:
| Facility | Location | Investment | Status |
|---|---|---|---|
| Biologics Manufacturing | Wilson, NC | $2B+ | Broke ground March 2025 |
| Cell Therapy | Lower Gwynedd, PA | $1B+ | Announced Feb. 2026 |
| Drug Product Manufacturing | North Carolina | TBD | Planned |
| Fujifilm CDMO Partnership | Holly Springs, NC | $2B (capacity deal) | Secured |
J&J already has ten facilities in Pennsylvania spanning more than 2 million square feet, with an estimated $10 billion annual economic impact across the state.
Carvykti: The Blockbuster Behind the Build
The Pennsylvania facility will expand production capacity for cell therapies targeting cancer, immune-mediated diseases, and neurological conditions. While J&J hasn't specified which products will be manufactured at the site, the investment timing aligns with the explosive commercial trajectory of Carvykti, the CAR-T cell therapy J&J co-developed with Legend Biotech.
Carvykti sales nearly doubled in 2025, reaching $1.9 billion (up 96% year-over-year) as the therapy became the leading CAR-T in multiple myeloma with more than 10,000 patients treated across 14 markets.
"Carvykti is the leading CAR-T cell therapy in multiple myeloma, with more than 10,000 patients now treated across 14 markets, and we are not stopping there," CEO Joaquin Duato said on J&J's Q4 2025 earnings call.
The therapy's commercial success has been constrained by manufacturing capacity — a common bottleneck for personalized CAR-T treatments that require extracting, engineering, and reinfusing each patient's own T cells. J&J and Legend have been aggressively expanding production, including:
- Raritan, NJ expansion: Physical capacity expansion expected to receive approval by end of 2025
- Tech Lane facility (Ghent, Belgium): Commercial production initiated in late 2025 for European market
- Novartis partnership: Commercial manufacturing contract initiated in January 2025
Legend Biotech's manufacturing success rate stands at 97% — "the highest in the CAR-T industry" — with a 30-day turnaround time from cell collection to infusion.
Financial Strength Supports Capex Buildout
J&J's balance sheet comfortably supports the multi-billion-dollar domestic manufacturing expansion. The company generated $94.2 billion in revenue in FY 2025, up 6% year-over-year, with $4.8 billion in capital expenditures.
| Metric | FY 2022 | FY 2023 | FY 2024 | FY 2025 |
|---|---|---|---|---|
| Revenue ($B) | $80.0 | $85.2 | $88.8 | $94.2 |
| Gross Margin % | 69.3% | 69.2% | 69.6% | 68.1% |
| EBIT Margin % | 27.7% | 27.8% | 25.4% | 28.0% |
| Capital Expenditure ($B) | $3.7 | $4.4 | $4.4 | $4.8 |
The Innovative Medicine segment — which houses Carvykti — delivered $60.4 billion in sales for 2025, up 6% operationally, led by oncology ($25.4 billion, +22%) and the continued growth of Tremfya ($5.2 billion, +41%).
What to Watch
Near-term catalysts:
- Carvykti frontline data: CARTITUDE-5 and CARTITUDE-6 trials are fully enrolled, targeting first-line multiple myeloma — a significantly larger patient population
- Manufacturing milestones: Raritan expansion approval expected by year-end; continued European capacity ramp
- Additional tariff developments: The White House has signaled ongoing negotiations with other pharma companies, which could reshape competitive dynamics
Risks:
- Patent litigation: 2seventy bio filed suit in January 2026 alleging Carvykti infringes its EU patents; J&J faces potential damages and injunction risk in Europe
- Competition: Bristol Myers Squibb's Breyanzi and Abecma, along with bispecific antibodies, continue to compete for multiple myeloma market share
- Execution: Cell therapy manufacturing is complex and capital-intensive; delays or quality issues could constrain growth
Related Companies:
- Johnson & Johnson — Global healthcare company; Carvykti co-developer
- Legend Biotech — CAR-T cell therapy developer; Carvykti partner
- Novartis — Contract manufacturer for Carvykti