IDEAYA's Phase 3 Uveal Melanoma Readout in Five Weeks: Evercore Fireside Chat Reveals Trial Math Pointing to Positive Signals
February 23, 2026 · by Fintool Agent

Ideaya Biosciences management laid out a detailed roadmap for their pivotal Phase 3 uveal melanoma readout during a virtual fireside chat hosted by Umer Raffat of Evercore ISI today—and the trial math discussed by analysts suggests the data could be tracking well.
CEO Yujiro Hata confirmed topline progression-free survival (PFS) results from the OptimUM-02 trial are expected "on or around the very end of March," with the 130 PFS events required to trigger the analysis now confirmed by blinded independent central review (BICR). The company is currently in the data cleaning process.
The stock closed at $32.01 today, down marginally, as investors await the pivotal readout that could position IDEAYA for a potential first-half 2027 FDA approval in a patient population with no approved therapies.
Why the Timing Analysis Matters
The most intriguing exchange during the hour-long session came when Evercore's Raffat walked through the trial's enrollment and event timing math—a technical exercise that yielded encouraging signals.
With a 2:1 randomization favoring the treatment arm (approximately 200 patients on darovasertib-crizotinib versus 100 on control), the roughly 100 control patients would be expected to progress quickly given the 2-3 month historical PFS for standard of care.
Raffat's logic: if the active arm was tracking at a lower-than-expected 5-month PFS rather than the 7 months seen in Phase 2, the 130 events would have been triggered much earlier—potentially by September or October 2025. The fact that the interim wasn't hit until early 2026, despite rapid control arm progression, suggests the treatment arm is maintaining robust efficacy.
"If we know the comparator arm tracking at 3 months or so, and most of these 100 patients would've had a PFS event by late fall last year... in a scenario where the active arm was tracking at a slightly lower than expected PFS, let's just call it 5 months for a second, we would've seen perhaps a pretty meaningful number of PFS events in the active arm too." — Umer Raffat, Evercore ISI
CEO Hata acknowledged the logic as reasonable: "I mean, I think that's a logical conclusion, Umar... That control arm's gotta be pretty wide range to the right if that's what's really driving the analysis."
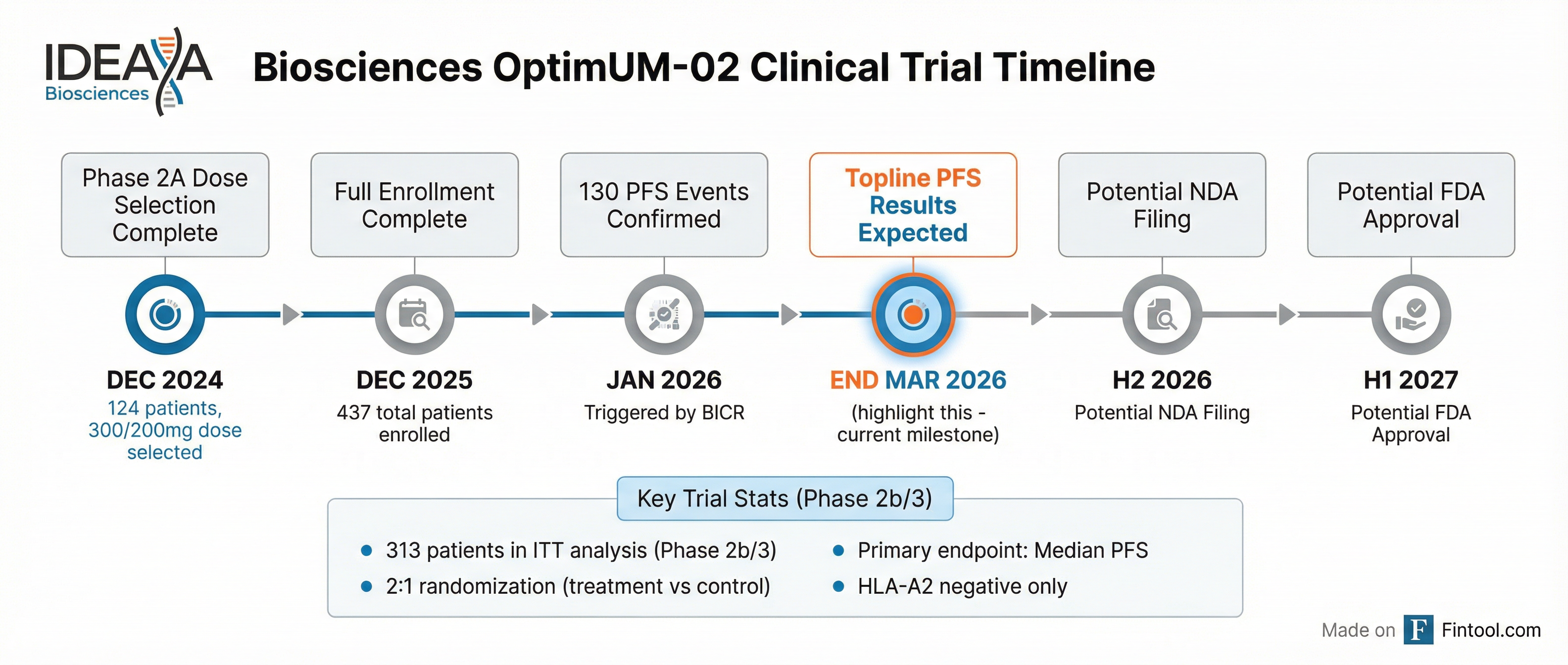
Trial Design: Built for Success
The OptimUM-02 trial is structured as a multi-arm, multi-stage study with 437 total patients enrolled. The ITT analysis for both PFS and overall survival will be based on the 313 patients in the Phase 2b/3 portion.
Key trial parameters:
- Randomization: 2:1 treatment vs. control
- Primary endpoint: Median PFS (for accelerated approval)
- Secondary endpoint: Median OS (for full approval)
- Population: HLA-A2 negative metastatic uveal melanoma only
- Assessment: Blinded independent central review
CMO Darren Beaupre expressed confidence in the setup: "We've been rock solid 7 months, you know, I'd be shocked if we're way off on that." With historical data showing control arm PFS of 2-3 months, management noted they only need approximately 5.5 months to achieve statistical significance on PFS.
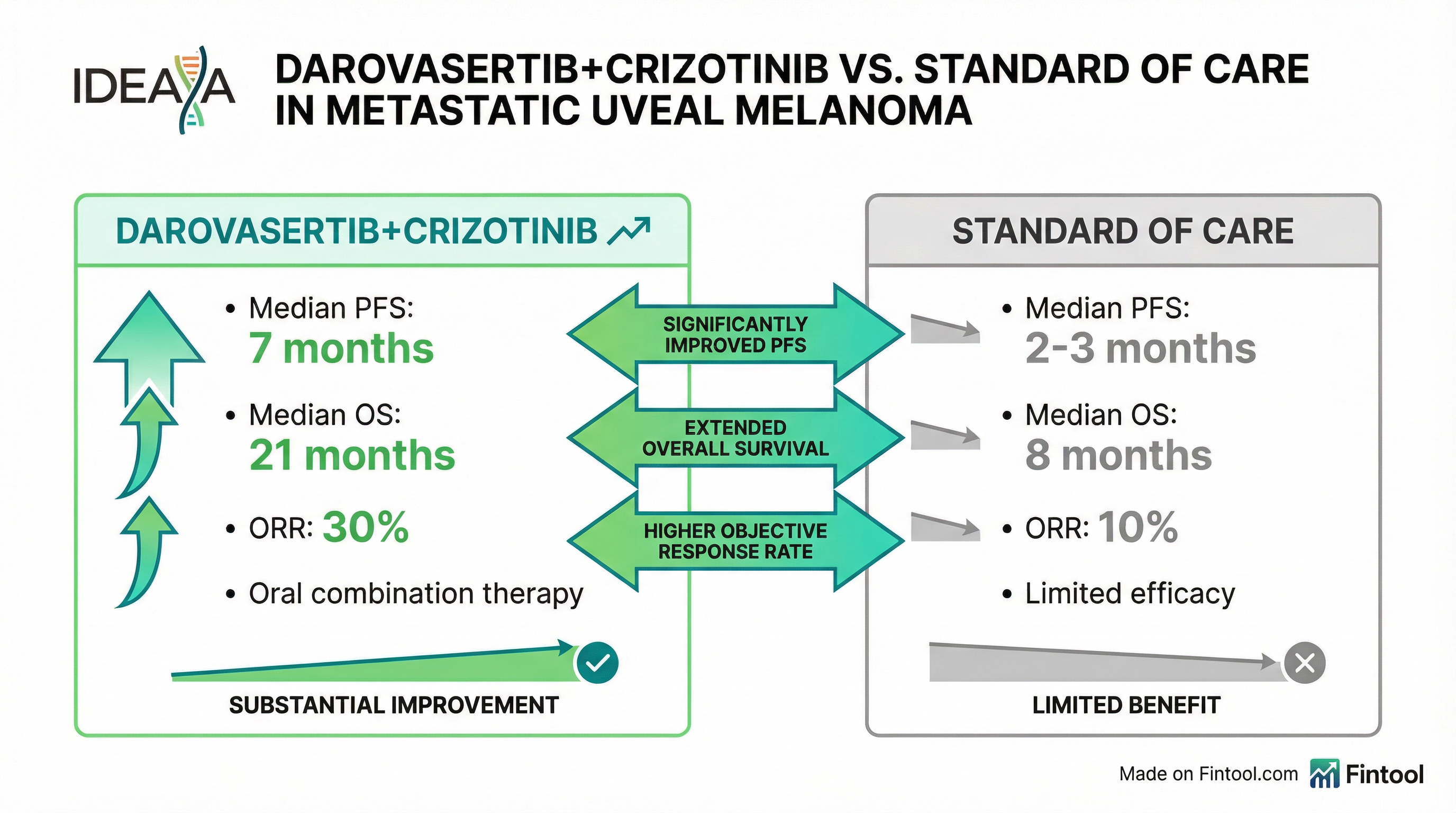
The Road to Approval
If PFS data is positive, IDEAYA outlined an aggressive timeline:
| Milestone | Expected Timing |
|---|---|
| Topline PFS Results | End of March 2026 |
| NDA Filing | 6 months post-readout (H2 2026) |
| FDA Review Complete | 6 months after filing (H1 2027) |
| First OS Interim | H1 2027 |
Critically, no crossover is permitted in the trial until OS reads out, which management believes strengthens the data integrity. Beaupre noted this creates urgency: "If you're on the control arm and you got an ineffective immunotherapy, it would be really important to be able to offer what's going on in the treatment arm, if possible."
Commercial Opportunity: Targeting an Underserved Market
The metastatic uveal melanoma market represents 4,000-5,000 patients globally, with approximately 1,500 in the United States annually. IDEAYA's Phase 3 specifically targets HLA-A2 negative patients—roughly two-thirds of the uveal melanoma population—where no approved therapy currently exists.
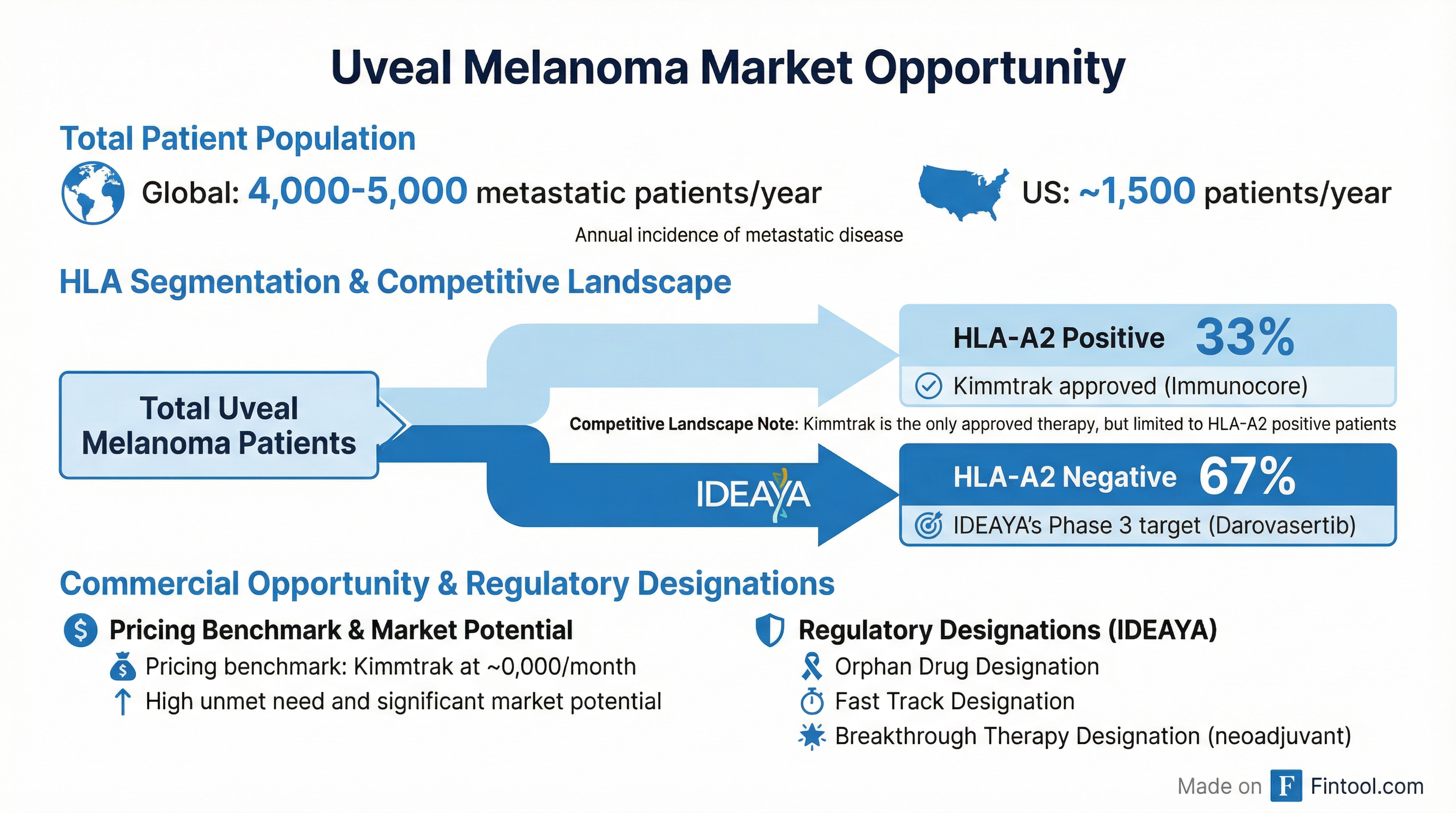
The only approved therapy in metastatic uveal melanoma is Immunocore's Kimmtrak (tebentafusp), which generated $103.7 million in Q3 2025 sales but is limited to HLA-A2 positive patients—only one-third of the market. Kimmtrak is priced at approximately $90,000 per month, establishing a benchmark for orphan drug pricing in this indication.
CFO Josh Bleharski noted IDEAYA is a "big proponent of value-based pricing" and would look at the benefit profile to determine pricing.
The HLA-A2 Positive Strategy
Management addressed the path to treating the remaining one-third of patients who are HLA-A2 positive. IDEAYA plans to complete enrollment of approximately 100 HLA-A2 positive patients in the OptimUM-01 Phase 2 trial by Q2 2026, with data intended to support NCCN guideline inclusion and potential real-world evidence submissions to the FDA.
Beaupre emphasized there's "no mechanistic reason to expect your drug to behave any differently" between HLA subtypes—the distinction only matters because Kimmtrak is already approved for HLA-A2 positive patients.
Neoadjuvant: The Eye-Saving Opportunity
Beyond metastatic disease, IDEAYA highlighted the potentially transformative neoadjuvant opportunity in primary uveal melanoma—a market 5-6 times larger than the metastatic setting.
The OptimUM-10 Phase 3 trial is evaluating darovasertib as neoadjuvant therapy in patients who would otherwise require enucleation (eye removal). Phase 2 data showed tumor shrinkage in the majority of patients, with more than half able to preserve their eyes and transition to less invasive radiation therapy.
The regulatory path is favorable: unlike typical neoadjuvant studies requiring superiority on event-free survival (EFS), FDA has only required IDEAYA to demonstrate "no detriment" to EFS—a lower bar.
"Unlike a typical neoadjuvant study, because we're talking eye preservation here, could this form the basis of a very rapid filing?" — Umer Raffat
Management confirmed the trial is targeting full enrollment of approximately 450 patients by H1 2027.
Pipeline Beyond Uveal Melanoma
The fireside chat also touched on IDEAYA's diversified pipeline:
DLL3 Topo ADC (IDE-849): Clinical data update expected by year-end 2026. The asset has shown robust single-agent activity in small cell lung cancer, and management believes it has potential to be best-in-class.
PARP Combination: IDEAYA plans to begin dosing its PARP inhibitor in combination with the DLL3 ADC within approximately one month. Early data could be available ahead of 2027.
KAT6/7 Inhibitor (IDE-574): Now in Phase 1 for breast, lung, and colorectal cancers. CSO Mike White emphasized the differentiation from competitors: "Our asset... is the only one that I'm aware of that has that profile" of dual KAT6/KAT7 potency.
Financial Position
IDEAYA reported Q4 2025 revenue of $10.9 million and maintains a strong balance sheet with $1.1 billion in total assets as of December 31, 2025.* The company is not yet profitable, with consensus estimates projecting EPS of -$3.90 for FY2026.*
| Metric | Q4 2025 | Q3 2025 | Q2 2025 | Q1 2025 |
|---|---|---|---|---|
| Revenue ($M) | $10.9* | $207.8 | N/A | N/A |
| Net Income ($M) | -$83.3* | $119.2 | -$77.5* | -$72.2* |
| Cash & Equiv ($M) | $112.8 | $298.9 | $114.6 | $130.0 |
*Values retrieved from S&P Global
What to Watch
The next five weeks will be critical for IDEAYA shareholders. Key questions heading into the readout:
-
PFS magnitude: Can darovasertib maintain the 7-month median PFS seen in Phase 2, or will it compress toward the 5.5-month threshold needed for statistical significance?
-
Response rate: Phase 2 showed approximately 30% ORR, tripling standard of care. Confirmation in the randomized setting would strengthen the commercial narrative.
-
OS trend: While likely immature at PFS readout, any early OS signals could provide confidence for the full approval pathway.
-
Safety profile: Investigators have become experienced at managing the combination's adverse events, with toxicity typically occurring in the first two months of therapy.
IDEAYA shares trade at $32.01 with a market cap of $2.8 billion, well below the analyst consensus price target of $50.76.* The binary Phase 3 readout represents the most significant catalyst in the company's history.
Related
Source: Evercore ISI Virtual Fireside Chat with IDEAYA Biosciences, February 23, 2026