Novo Nordisk's CagriSema Fails to Match Eli Lilly's Zepbound in Head-to-Head REDEFINE 4 Trial
February 23, 2026 · by Fintool Agent
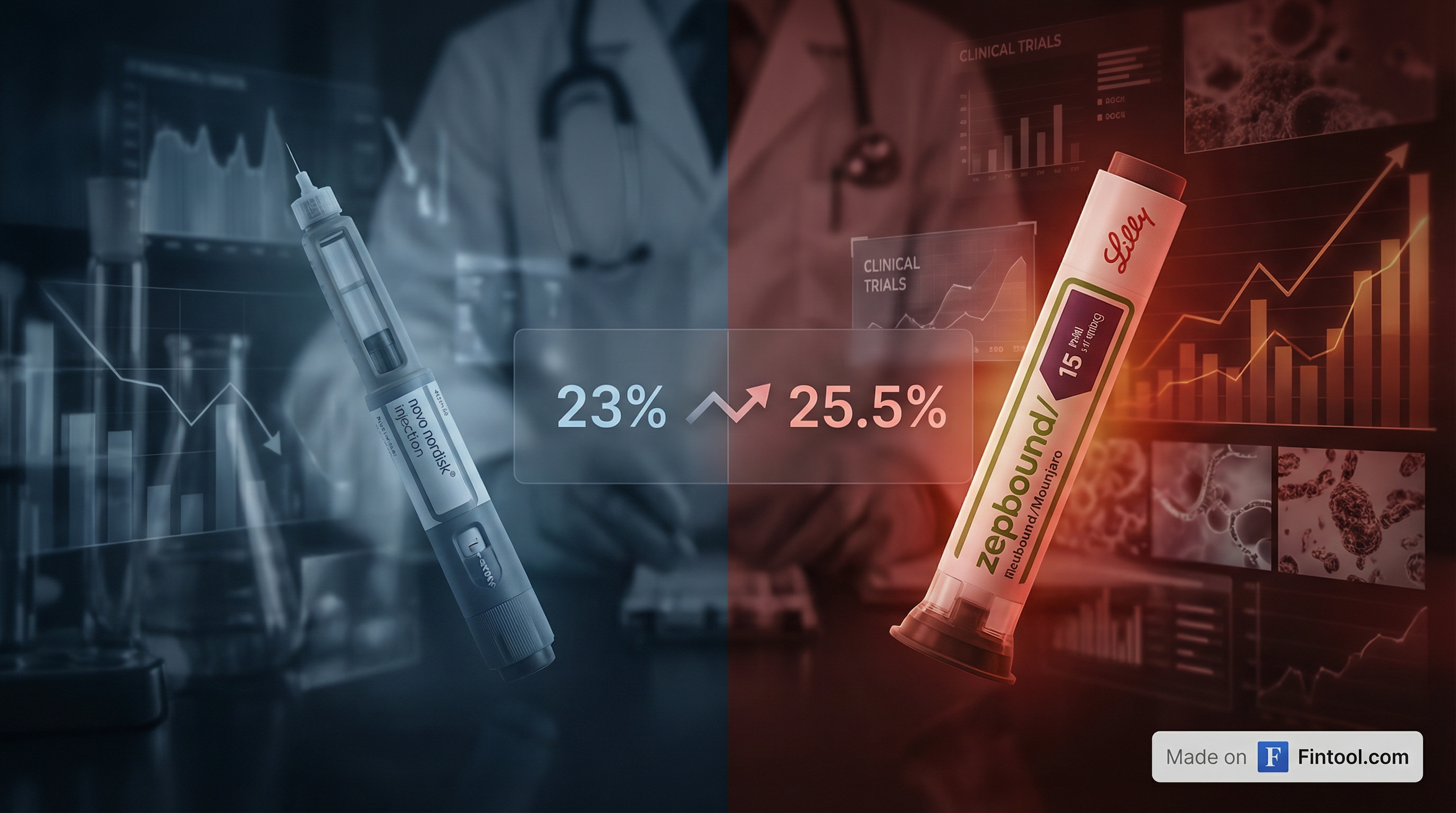
Novo Nordisk suffered a significant setback Monday in its battle against Eli Lilly for obesity drug supremacy, as its next-generation weight loss drug CagriSema failed to demonstrate non-inferiority against Lilly's tirzepatide in a head-to-head clinical trial.
The REDEFINE 4 trial results showed CagriSema achieved 23.0% weight loss after 84 weeks compared to 25.5% for tirzepatide—the active ingredient in Lilly's blockbuster Zepbound and Mounjaro. The 2.5 percentage point gap represents a meaningful difference in a market where efficacy is king.
Novo Nordisk shares collapsed approximately 15% in after-hours trading, falling to around $40.15 from Friday's close of $47.42. Eli Lilly shares rose about 3% to $1,040 in the premarket session.

The Trial: What Went Wrong
REDEFINE 4 enrolled 809 people with obesity and at least one comorbidity, with a mean baseline body weight of 114.2 kg (252 lbs). Both CagriSema and tirzepatide were administered as once-weekly subcutaneous injections over 84 weeks.
The trial employed an open-label design—meaning both investigators and participants knew which drug they were receiving—a factor Novo Nordisk's management quickly highlighted as a potential source of bias.
"More than 40% of the investigators are previous investigators on the comparator drug. They know the drug well. They have probably prescribed the drug, and they feel confident in the drug," said Martin Holst Lange, EVP of Research and Development at Novo Nordisk. "We know that drives the bias, and that's maybe why we have seen these surprisingly good data for the comparator drug."
The company also noted that fewer CagriSema patients reached the highest 2.4mg dose compared to tirzepatide patients reaching 15mg, potentially limiting efficacy.
A Pattern of Underperformance
Today's disappointment follows a similar miss in REDEFINE 1, announced in December 2024, where CagriSema achieved 22.7% weight loss—below management's 25% expectations. The pattern raises questions about whether CagriSema can truly compete with Lilly's tirzepatide franchise.
| Trial | Drug | Duration | Weight Loss | Primary Endpoint |
|---|---|---|---|---|
| REDEFINE 1 | CagriSema 2.4/2.4mg | 68 weeks | 22.7% | Met (vs placebo) |
| REDEFINE 4 | CagriSema 2.4/2.4mg | 84 weeks | 23.0% | Not Met (vs tirzepatide) |
| REDEFINE 4 | Tirzepatide 15mg | 84 weeks | 25.5% | — |
| SURMOUNT-1 | Tirzepatide 15mg | 72 weeks | 22.5% | Met (vs placebo) |
Notably, the 25.5% weight loss for tirzepatide in REDEFINE 4 exceeded its performance in Lilly's own SURMOUNT trials, a fact Novo management called "surprising" and attributed to the open-label design.
Market Reaction and Competitive Context
The setback couldn't come at a worse time for Novo Nordisk. The Danish drugmaker has been losing ground to Lilly throughout 2025 and into 2026:
Since January 2025:
- Novo Nordisk: Down 46% (from $87.52 to $47.42 before today's drop)
- Eli Lilly: Up 30% (from $778 to $1,010)
The divergence reflects a fundamental shift in market dynamics. Lilly's tirzepatide has captured an estimated 60% of the global branded GLP-1 market, with Novo holding approximately 39%. In the U.S., Novo estimates 7-8 out of 10 new patients choose Lilly's products.
| Metric | Novo Nordisk | Eli Lilly |
|---|---|---|
| 2026 Revenue Guidance | Decline 5-13%* | Growth 25%* |
| Market Cap (Feb 20) | $211B | $952B |
| GLP-1 Market Share | 39% | 60% |
| 2025 GLP-1 Revenue | $33B (Ozempic/Wegovy)* | $36.5B (Mounjaro/Zepbound)* |
*Values retrieved from S&P Global and news reports.
Management's Defense and Path Forward
Despite the miss, CEO Mike Doustdar struck a defiant tone on the investor call, arguing that labels—not head-to-head trial results—drive commercial success.
"The label that we have is, of course, based on not this trial, but the previous one, and the label that our competitor has also is below, basically, the CagriSema's label," Doustdar said. "We strongly believe that CagriSema has right now, the best weight efficacy than any product currently in the market."
Novo is pinning its hopes on several upcoming catalysts:
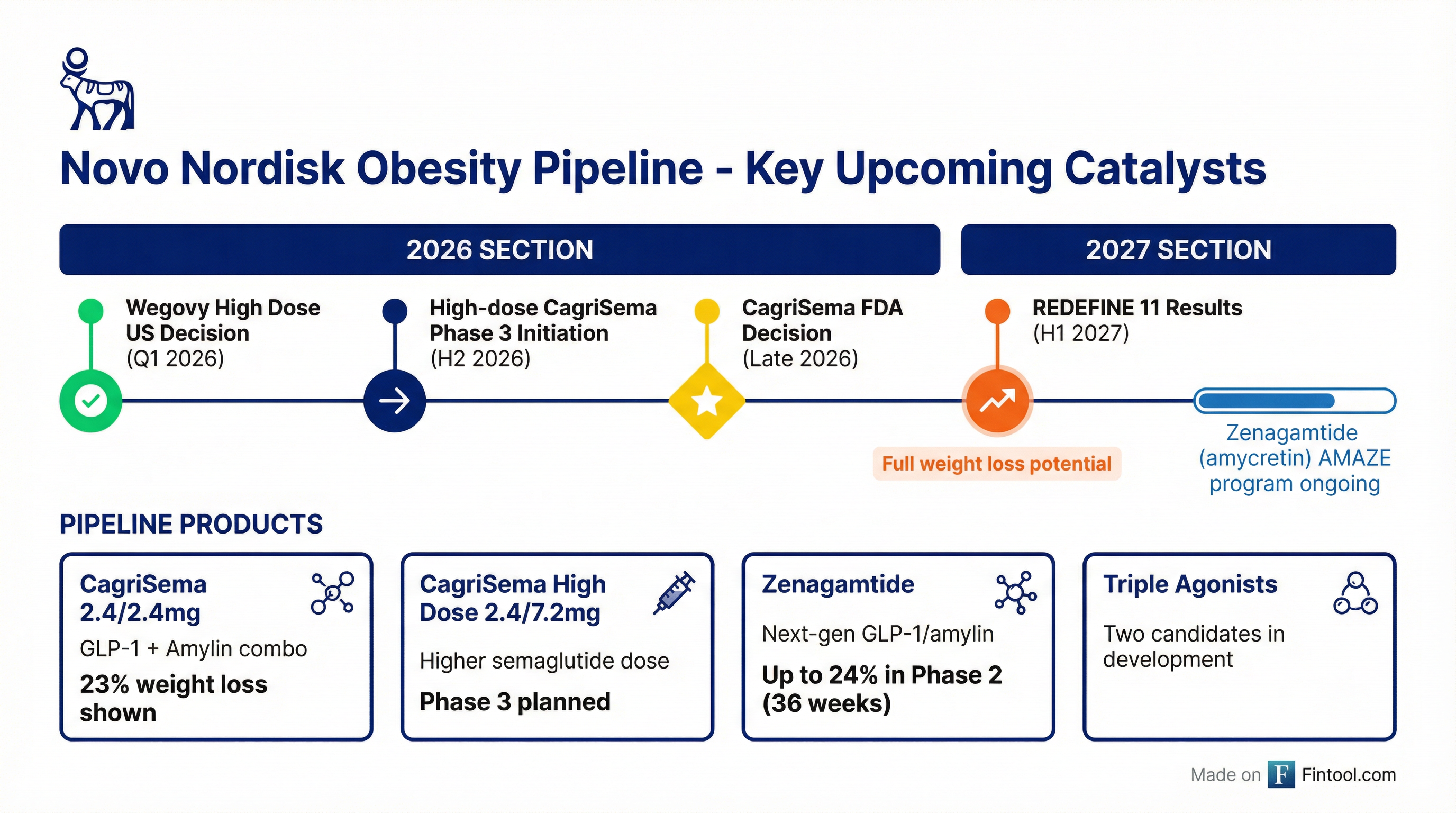
REDEFINE 11 (Results H1 2027): A redesigned trial incorporating learnings from REDEFINE 1 and 4, with longer duration and optimized dose titration protocols. Management believes this will unlock CagriSema's "full weight loss potential."
High-Dose CagriSema (Phase 3 initiation H2 2026): A new formulation combining 2.4mg cagrilintide with 7.2mg semaglutide (up from 2.4mg). Novo's STEP-UP trial showed 7.2mg semaglutide alone achieved 20.7% weight loss vs 17.5% for the 2.4mg dose.
Zenagamtide (amycretin) (AMAZE Phase 3 ongoing): Novo's next-generation GLP-1/amylin combination showed up to 24% weight loss after just 36 weeks in Phase 2—potentially superior efficacy in a shorter timeframe.
"I would not rule out that we would do additional head-to-head studies," Lange said when asked about future comparisons with tirzepatide.
The Broader Obesity War
The REDEFINE 4 results underscore Lilly's strengthening competitive position. Combined 2025 sales of Mounjaro and Zepbound reached $36.5 billion—accounting for 56% of Lilly's total revenue—making tirzepatide the world's best-selling drug.
Novo's semaglutide franchise (Ozempic + Wegovy) generated $33 billion in 2025, but the company warned in February that 2026 sales could decline 5-13% due to pricing pressure and patent expirations in several markets.
| NVO Financial Performance | Q1 2025 | Q2 2025 | Q3 2025 | Q4 2025 |
|---|---|---|---|---|
| Revenue ($B) | $11.3 | $12.1 | $11.8 | $12.4 |
| EBITDA Margin | 54.6% | 49.0% | 48.7% | 46.3% |
| Gross Margin | 83.5% | 83.3% | 80.7% | 82.1% |
*Values retrieved from S&P Global
The one bright spot for Novo: its Wegovy pill, launched in January 2026 as the first oral GLP-1 for obesity, reached 50,000 weekly prescriptions by late January and is priced at $149-299/month for cash payers. But Lilly's oral tirzepatide is expected to launch in Q2 2026, potentially eroding even that advantage.
What to Watch
Near-Term Catalysts:
- Q1 2026: Wegovy High Dose (7.2mg) FDA decision expected
- Late 2026: CagriSema FDA decision for obesity (based on REDEFINE 1/2)
- H2 2026: High-dose CagriSema Phase 3 trial initiation
Medium-Term:
- H1 2027: REDEFINE 11 results—critical for establishing CagriSema's true competitive position
- Ongoing: Zenagamtide (amycretin) AMAZE Phase 3 program
For investors, today's results suggest Novo Nordisk's path to regaining obesity market leadership runs through pipeline execution rather than current-generation products. The question is whether the market will be patient enough to wait—and whether Lilly's substantial head start can ever be closed.
Related: