Novo Nordisk's CagriSema Loses to Eli Lilly's Zepbound in Head-to-Head Trial, Stock Hits 5-Year Low
February 23, 2026 · by Fintool Agent
Novo Nordisk's next-generation obesity drug CagriSema failed to match Eli Lilly's blockbuster Zepbound in a head-to-head clinical trial, sending Novo shares plunging 15% to their lowest level since June 2021. The Danish pharma's stock hit $40.16 in U.S. trading, erasing more than $400 billion in market value since its 2024 peak.
The setback cements Lilly's dominance in the $100 billion-plus obesity drug market and deals a major blow to Novo's strategy of using CagriSema to reclaim market leadership from its Indianapolis-based rival.
The Trial That Changed Everything
In the REDEFINE 4 trial—an 84-week, open-label study of 809 patients with obesity—CagriSema achieved a 23.0% reduction in body weight compared to 25.5% for Lilly's tirzepatide. The trial's primary endpoint of demonstrating "non-inferiority" was not met, meaning CagriSema proved statistically inferior to Zepbound.

The 2.5 percentage point gap may sound small, but in the winner-take-all obesity market, it represents a significant competitive disadvantage.
"This is a worst-case scenario for Novo," said Markus Manns at Union Investment, a shareholder of both companies. "Now it is clinically proven that Mounjaro is better than CagriSema. The base case was that Mounjaro and CagriSema are similar... nobody had on the agenda that CagriSema would be worse."
Novo's Defense: "More to Come"
Novo's Chief Scientific Officer Martin Holst Lange attempted to explain the shortfall, pointing to several factors: the open-label trial design may have introduced bias toward the "well-established, well-known" Lilly drug, and CagriSema performed consistently with prior REDEFINE 1 results while the comparator "performed unusually well on efficacy."
CEO Mike Doustdar pushed back against the narrative that CagriSema is obsolete: "To say it's obsolete is quite belittling a fantastic drug. When CagriSema will make it to the market early next year as the first amylin-based product, it will have the best weight loss label than any marketed product at that time."
But the market wasn't buying it. Lilly shares rose 4% in pre-market trading.
| Metric | CagriSema | Tirzepatide (Zepbound) |
|---|---|---|
| Weight Loss (adherent) | 23.0% | 25.5% |
| Weight Loss (all patients) | 20.2% | 23.6% |
| Trial Duration | 84 weeks | 84 weeks |
| Sample Size | 400 patients | 400 patients |
| Primary Endpoint | Not met (non-inferiority) | Winner |
Source: Novo Nordisk REDEFINE 4 press release
The Great Divergence: A Tale of Two Stocks
The market has been voting with its feet for over a year. Since January 2024, Novo Nordisk shares have plummeted 61% while Eli Lilly has surged 78%—a staggering 139 percentage point divergence in the GLP-1 drug war.
Lilly's Zepbound has captured nearly 70% of new U.S. GLP-1 prescriptions in the branded obesity market, with Mounjaro commanding over 55% of the type 2 diabetes incretin market. In Q4 2025 alone, Zepbound generated $4.3 billion in revenue (+123% YoY) while Mounjaro brought in $7.4 billion (+110% YoY).
| Revenue Comparison | FY 2025 | YoY Growth |
|---|---|---|
| Eli Lilly | ||
| Mounjaro (diabetes) | $22.97B | +99% |
| Zepbound (obesity) | $13.54B | +175% |
| Total tirzepatide | $36.5B | +122% |
| Novo Nordisk | ||
| Total Revenue | $48.6B | +16% |
Sources: Eli Lilly Q4 2025 8-K , Novo Nordisk financials
Novo's Pipeline: Hope After CagriSema
Despite the setback, Novo isn't giving up. The company has an extensive pipeline of obesity drugs designed to eventually outperform even today's results.
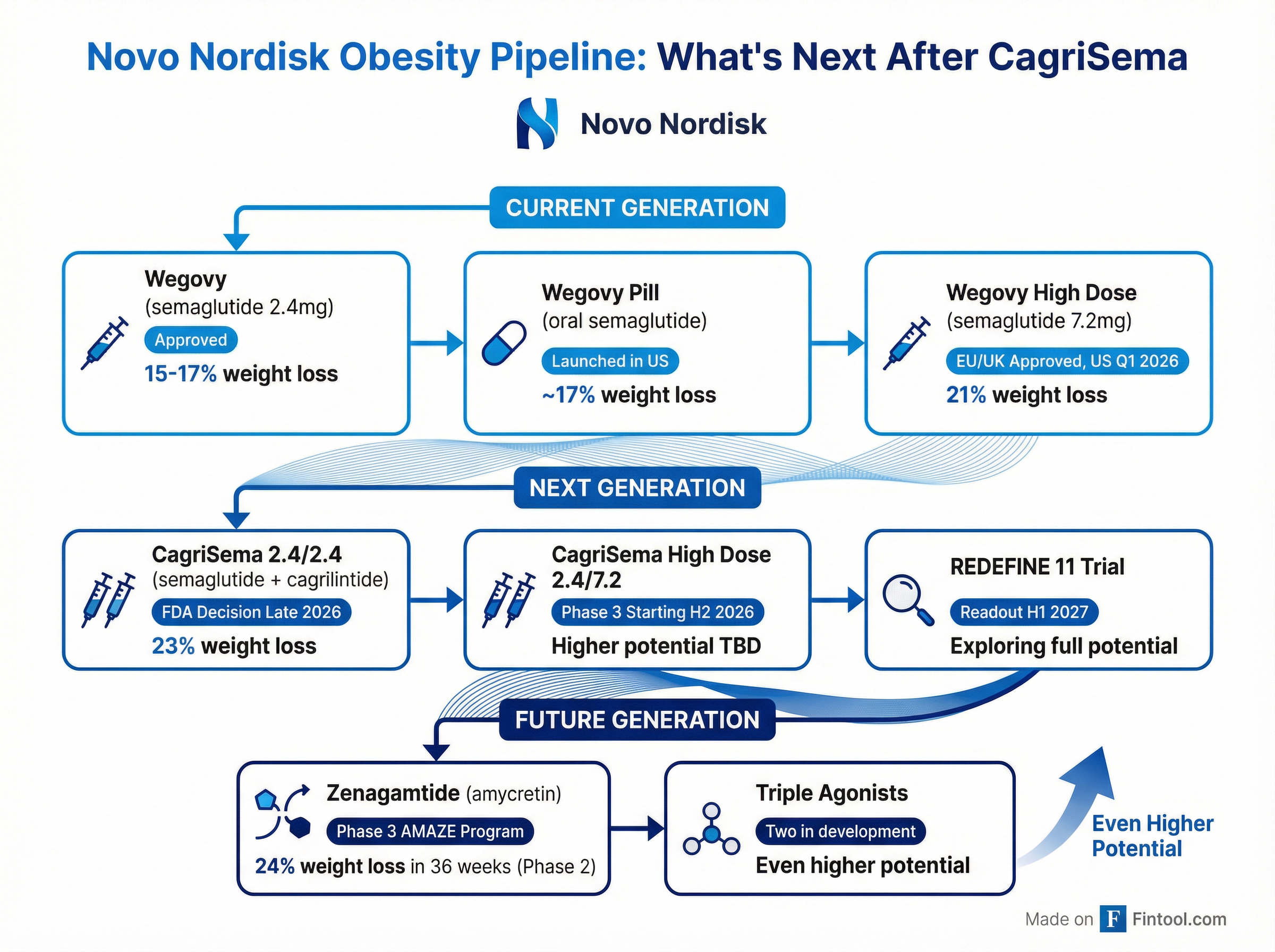
Key pipeline catalysts:
- CagriSema approval: FDA decision expected late 2026, with potential launch in early 2027
- REDEFINE 11 trial: Designed to show CagriSema's "full weight loss potential" with optimized dosing; readout H1 2027
- CagriSema High Dose (2.4mg/7.2mg): Phase 3 trial starting H2 2026 with triple the semaglutide dose
- Zenagamtide (amycretin): Next-generation GLP-1/amylin combo showed 24% weight loss in just 36 weeks in Phase 2; Phase 3 AMAZE program initiated
- Triple agonists: Two candidates in clinical development with potential for even higher weight loss
"We have multiple shots on goal," Doustdar emphasized. "Stand by for the results of the triple agonist that we will basically announce in due course. We have two triple agonists, and we are incredibly excited about that as well."
What to Watch
For Novo Nordisk:
- FDA decision on CagriSema (late 2026)
- Wegovy High Dose U.S. approval (Q1 2026 expected)
- REDEFINE 11 readout (H1 2027)
- Oral Wegovy launch momentum
- Zenagamtide Phase 3 progress
For Eli Lilly:
- Orforglipron (oral GLP-1) FDA decision expected Q2 2026
- Medicare obesity coverage implementation (July 1, 2026)
- Manufacturing capacity expansion
- International Zepbound launches
The Bigger Picture: The obesity drug market is projected to exceed $100 billion by 2030. Despite today's setback, both companies will likely capture enormous value as penetration remains in low-single digits. The question is whether Novo can regain its footing—or whether Lilly's lead becomes insurmountable.