Novo Nordisk Sues Hims & Hers for Patent Infringement as FDA Cracks Down on GLP-1 Compounders
February 9, 2026 · by Fintool Agent
Novo Nordisk filed a federal lawsuit against Hims & Hers Health on Monday, alleging patent infringement over compounded versions of its blockbuster weight-loss drugs Wegovy and Ozempic. The lawsuit caps a turbulent week that saw Hims launch—and then abruptly pull—a $49 compounded semaglutide pill after the FDA announced a broad crackdown on GLP-1 compounders.
HIMS shares crashed 25% Monday to $17.67, while NVO jumped nearly 6% to $49.46, nearly erasing last week's losses.
The Lawsuit: Seeking a Permanent Ban
Novo filed the lawsuit in U.S. District Court in Delaware, alleging Hims infringed US Patent 8,129,343 through its compounded semaglutide products. The Danish pharma giant is seeking a permanent injunction against Hims selling unapproved compounded drugs and is pursuing damages.
"Hims & Hers is mass marketing unapproved knock-off versions of Wegovy and Ozempic that evade the FDA's gold standard review process—that's dangerous and deceptive to patients, and undermines the scientific innovation and regulatory rigor in place to ensure these treatments are safe and effective," said John Kuckelman, Novo's senior vice president and group general counsel.
The lawsuit covers both oral and injectable compounded semaglutide products. Novo claims its testing found that injectable semaglutide drugs compounded by pharmacies contained impurities of up to 86%, with compounded oral versions containing impurities as high as 75%.
Hims fired back, calling the lawsuit "a blatant attack by a Danish company on millions of Americans who rely on compounded medications for access to personalized care."
"Once again, Big Pharma is weaponizing the U.S. judicial system to limit consumer choice," Hims said in a statement.
From Partnership to Litigation: A Dramatic Reversal
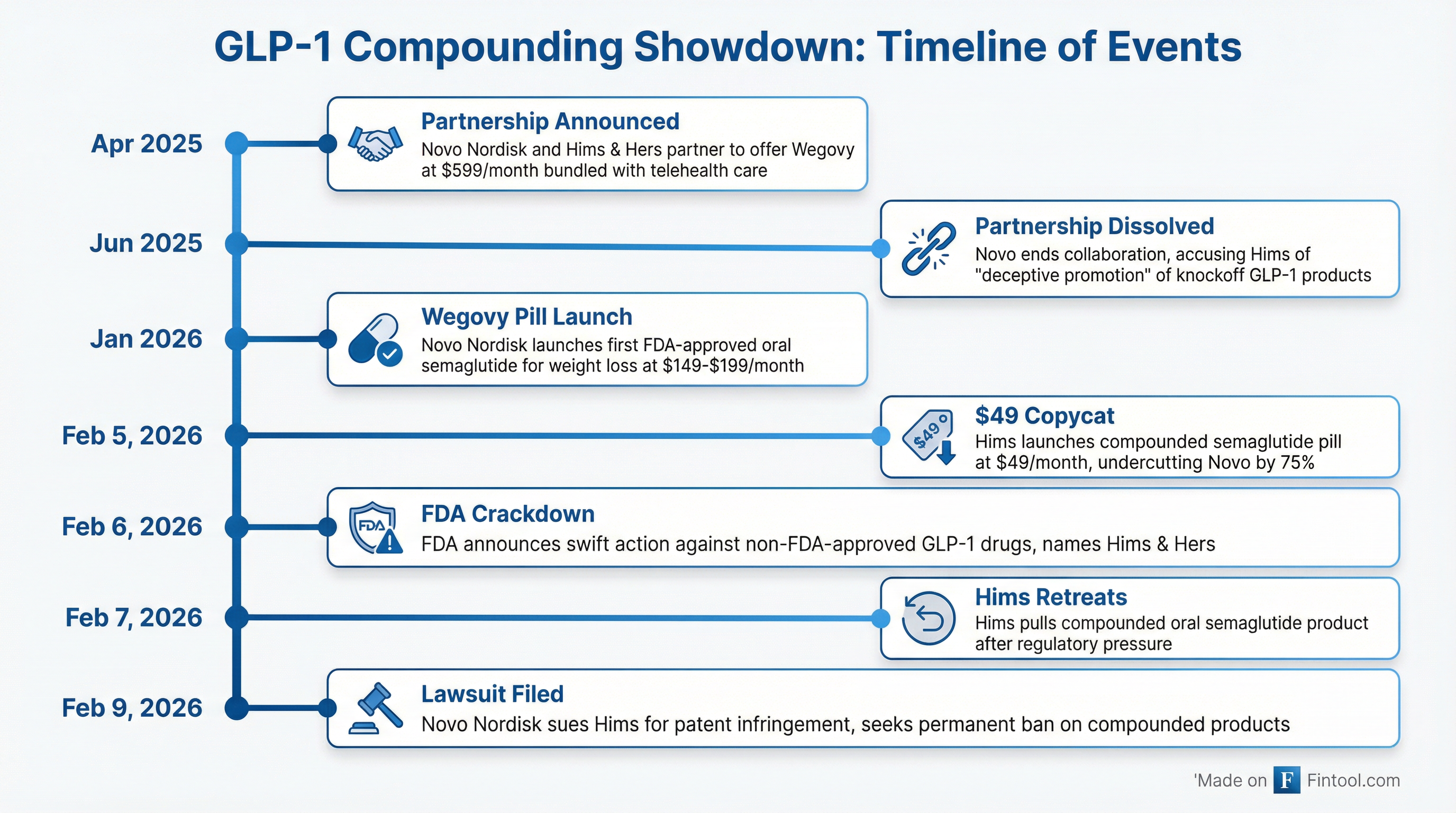
The lawsuit marks a dramatic escalation in what was once a collaborative relationship. In April 2025, Novo and Hims announced a partnership to make "proven obesity care and treatments more accessible and affordable," offering bundled access to Wegovy for $599 per month.
That partnership dissolved just two months later when Novo accused Hims of "deceptive promotion and selling of illegitimate, knockoff versions of Wegovy that put patient safety at risk." Hims CEO Andrew Dudum countered that Novo was attempting to control how clinicians at Hims make prescribing decisions.
The $49 Pill That Sparked a Firestorm
The immediate catalyst was Hims' February 5 launch of a compounded oral semaglutide pill priced at $49 per month—roughly $100 less than Novo's Wegovy pill, which launched in January at $149 for first-time users and $199 after.
The announcement sent shockwaves through the market. Novo shares hit their lowest level since July 2021 on February 5, while Eli Lilly—which is expected to launch its own GLP-1 pill in April—also sold off on fears of pricing pressure.
Novo CEO Mike Doustdar dismissed the Hims product in a meeting with investors: "People spending $49 on a Hims' pill would be wasting their money... if you swallow my injection, either in a liquid format or a pill format, your gut enzyme will basically get rid of it, and it will not get to your bloodstream. I have no idea what they have done with the compound. But assuming you just take the compound without the things around it and formulations and what have you, then this just doesn't work."
Novo's Wegovy pill uses proprietary SNAC technology to facilitate semaglutide absorption when administered orally—technology protected by formulation patents extending "into the late 30s," according to Doustdar.
FDA Crackdown: A "Tipping Point"
The FDA's response was swift and decisive. On February 6, FDA Commissioner Marty Makary announced the agency would move against companies mass-marketing unapproved GLP-1 drugs, explicitly naming Hims & Hers.
"These actions are aimed to safeguard consumers from drugs for which the FDA cannot verify quality, safety, or efficacy," the FDA stated. "We take seriously any potential violations of the Federal Food, Drug, and Cosmetic Act."
Hims pulled its compounded oral semaglutide product on February 7, just two days after launch.
Analysts see the FDA action as a pivotal shift. "They are not only declaring war on Hims & Hers' Wegovy pill, but GLP-1 compounders in general," said Sydbank analyst Soren Lontoft Hansen.
Novo's Kuckelman described the Hims pill launch as a "tipping point" that prompted coordinated action.
Financial Stakes for Hims
The lawsuit threatens a significant revenue stream for Hims. Leerink Partners estimates that Hims' weight loss business represents approximately $800 million in fiscal year 2025 revenue, operating at a low double-digit EBITDA margin. This segment accounts for roughly 25-30% of the company's total EBITDA.
| Metric | Q4 2024 | Q1 2025 | Q2 2025 | Q3 2025 |
|---|---|---|---|---|
| Revenue ($M) | $481.1 | $586.0 | $544.8 | $599.0 |
| Gross Margin | 76.8% | 73.5% | 76.4% | 73.8% |
| EBITDA Margin | 6.0% | 11.5% | 6.8% | 4.9% |
Leerink cut its price target on HIMS to $20 from $24, reducing its valuation multiple to approximately 10x calendar year 2026 EBITDA from 13.5x previously, "reflecting the increased legal uncertainty and fundamental risk facing the company's business model."
Hims disclosed the regulatory risks in its Q3 2025 10-Q: "Resolution of the shortage has constrained and is expected to continue to constrain our ability to continue providing access to compounded semaglutide on our platform."
The Compounding Loophole
Compounded drugs have proliferated in the GLP-1 market due to a regulatory loophole. When FDA-approved drugs face shortages, compounding pharmacies can legally produce bulk copies. Semaglutide experienced shortages throughout 2024 and early 2025, creating an opening for compounders.
The FDA declared the semaglutide shortage resolved on February 21, 2025, and the enforcement discretion period for 503B outsourcing facilities ended on May 22, 2025. Despite this, compounding has "illicitly continued," according to Novo's 20-F filing.
Hims currently uses 503A compounding pharmacies for GLP-1 fulfillment, which operate under different rules than the larger 503B outsourcing facilities. The company acquired MedisourceRx, a licensed 503B compounding facility, in September 2024, but its current use is limited.
The regulatory landscape continues to evolve. Novo noted in its 20-F that manufacturers have requested the FDA add semaglutide and tirzepatide to its "Demonstrable Difficulties for Compounding List"—a move that, if finalized, would effectively ban compounding of these drugs entirely.
Broader Implications for the GLP-1 Market
The lawsuit and FDA crackdown could reshape the competitive dynamics in the $50+ billion GLP-1 market.
| Company | Q4 2025 Revenue ($B) | Key Product | Status |
|---|---|---|---|
| Novo Nordisk | $12.4* | Wegovy, Ozempic | Lawsuit filed, FDA support |
| Eli Lilly | $15.5* | Zepbound, Mounjaro | Pill launch expected April 2026 |
| Hims & Hers | $0.6 | Compounded semaglutide | Under legal pressure |
*Values retrieved from S&P Global
Novo has been losing market share to both Eli Lilly and compounders. Its 20-F notes that Ozempic's position as the leading GLP-1 for type 2 diabetes "was overtaken by a competing GLP-1 based product during 2025."
However, the FDA action could provide relief. "I think the FDA made it quite clear that they would not tolerate a compounded Wegovy pill. This was an attack on the authority of the FDA," said Markus Manns at Union Investment, a Novo and Lilly shareholder.
Both Novo and Lilly have struck pricing deals with the Trump Administration, and their weight-loss drugs are featured on the new TrumpRx discounted-drugs website.
What to Watch
Legal timeline: The lawsuit is in its early stages. Key milestones include Hims' response, potential preliminary injunction hearings, and whether Hims will be permitted to continue selling compounded injectable semaglutide during trial.
FDA enforcement: Commissioner Makary has signaled aggressive use of "all available compliance and enforcement tools." Watch for warning letters, seizures, or injunctions against other compounders.
Hims' strategic response: The company maintains it has "a long history of providing safe access to personalized healthcare." Investors will watch whether Hims pivots its business model or contests the regulatory interpretation.
Eli Lilly's April launch: Lilly's oral GLP-1 launch will test whether legitimate competition can coexist with branded products at sustainable price points.