Ocular Therapeutix Makes Wet AMD History With AXPAXLI Superiority Win, Yet Stock Crashes 28%
February 17, 2026 · by Fintool Agent

Ocular Therapeutix achieved what no biotech company has accomplished in over two decades—demonstrating superiority to an approved anti-VEGF treatment in wet age-related macular degeneration—yet shares crashed 28% as the control arm's unexpectedly strong performance clouded the commercial thesis.
In a conference call this morning announcing SOL-1 Phase 3 results, CEO Dr. Pravin Dugel declared it "a historic day for Ocular Therapeutix, for the global retina community, and most importantly, for patients suffering from wet AMD."
The market disagreed. OCUL closed at $6.39, down from $8.88 the prior session, erasing roughly $540 million in market value as investors questioned whether the magnitude of differentiation justifies AXPAXLI's commercial potential in a $10+ billion market dominated by Regeneron's Eylea franchise.
The Historic Achievement
AXPAXLI became the first and only investigational product with a novel mechanism of action to demonstrate superiority to an approved anti-VEGF agent in a Phase 3 FDA-aligned trial—a bar that has "eluded the field for more than two decades," according to management.
The primary superiority endpoint was met with high statistical significance:
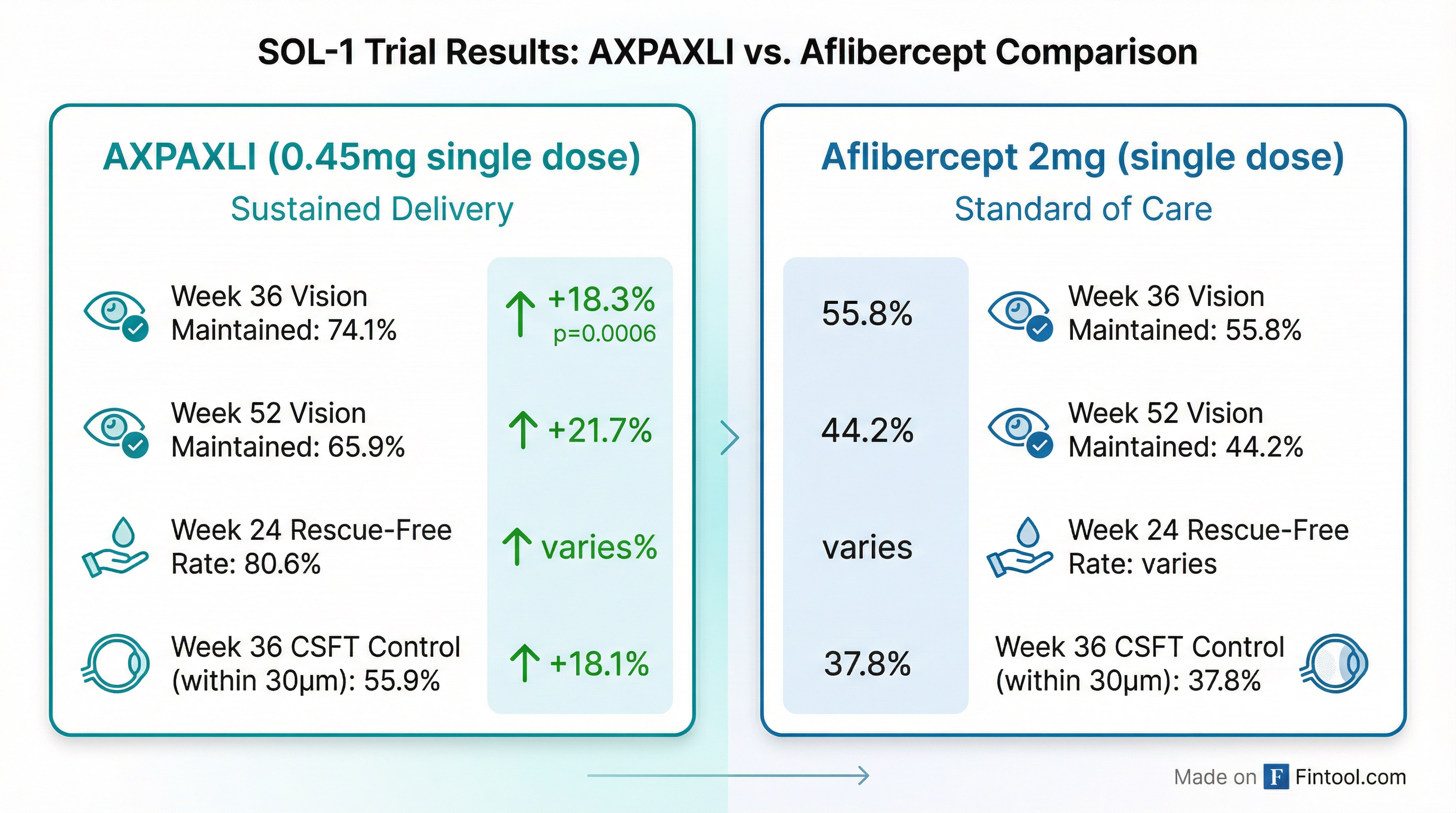
| Endpoint | AXPAXLI (0.45mg) | Aflibercept (2mg) | Risk Difference | P-Value |
|---|---|---|---|---|
| Vision Maintained Week 36 (Primary) | 74.1% | 55.8% | 17.5% | 0.0006 |
| Vision Maintained Week 52 | 65.9% | 44.2% | 21.1% | <0.0001 |
| CSFT Within 30μm Week 36 | 55.9% | 37.8% | 17.1% | 0.0013 (nominal) |
| Rescue-Free Week 24 | 80.6% | — | — | — |
| Rescue-Free Week 52 | 68.8% | 47.7% | — | — |
*Source: SOL-1 Top Line Data Conference Call *
"Let me repeat," Dugel emphasized during the call. "AXPAXLI is now the first and only investigational product in wet AMD with a novel mechanism of action to successfully demonstrate superiority to an approved anti-VEGF agent since their initial approval in wet AMD more than 20 years ago. Many programs have tried to reach this bar. All have failed. Today, AXPAXLI succeeded."
The Control Arm Paradox
The selloff centers on aflibercept's better-than-anticipated performance. At Week 36, 55.8% of patients receiving a single aflibercept injection maintained vision—far above what investors modeled for a comparator receiving essentially no active treatment for nine months.
Dr. Darius Moshfeghi, Chief of the Retina Division at Stanford's Byers Eye Institute and a rescue monitor in the trial, addressed the elephant in the room directly: "Why did the control arm do so well? I mean, it's really remarkable that the control arm did that well, much better than anyone expected, and yet AXPAXLI still beat the superiority outcome."
Management was defensive. Dugel called the control arm discussion "a purely academic exercise" with "absolutely no practical or clinical relevance whatsoever," arguing no physician would adopt a treat-and-observe approach based on these results.
But the commercial implications are harder to dismiss. STAT News reported the durability difference was "narrower than investors expected," sparking debate about AXPAXLI's competitive positioning.
The Clinical Case for Adoption
Despite market skepticism, the physicians on the call made a compelling case for immediate adoption if approved.
Dr. Arshad Khanani, Steering Committee Chair for the SOL program, emphasized the anatomical data: "55.9% of patients treated with AXPAXLI maintained central subfield thickness within 30 microns from baseline at week 36... We know that CSFT fluctuations of 50 microns or more lead to irreversible vision loss over time."
The rescue-free rates were particularly striking. At Week 24, 80.6% of AXPAXLI patients remained rescue-free under SOL-1's stringent criteria. When applying the more clinically relevant SOLR criteria (>5 letter loss plus >75μm CSFT increase), approximately 77% would have remained rescue-free—a proxy for how patients would fare in real-world practice.
"If AXPAXLI were available tomorrow, I believe retinal physicians would use it immediately and broadly in their patients with wet AMD," Khanani stated.
Dr. Moshfeghi concurred: "AXPAXLI's SOL-1 performance is exactly the type of product profile that I would expect to see rapid day one adoption, if approved. In my view, AXPAXLI represents the most consequential advance in wet AMD since the approval of the first anti-VEGF therapy over 20 years ago."
Regulatory Path and Next Catalysts
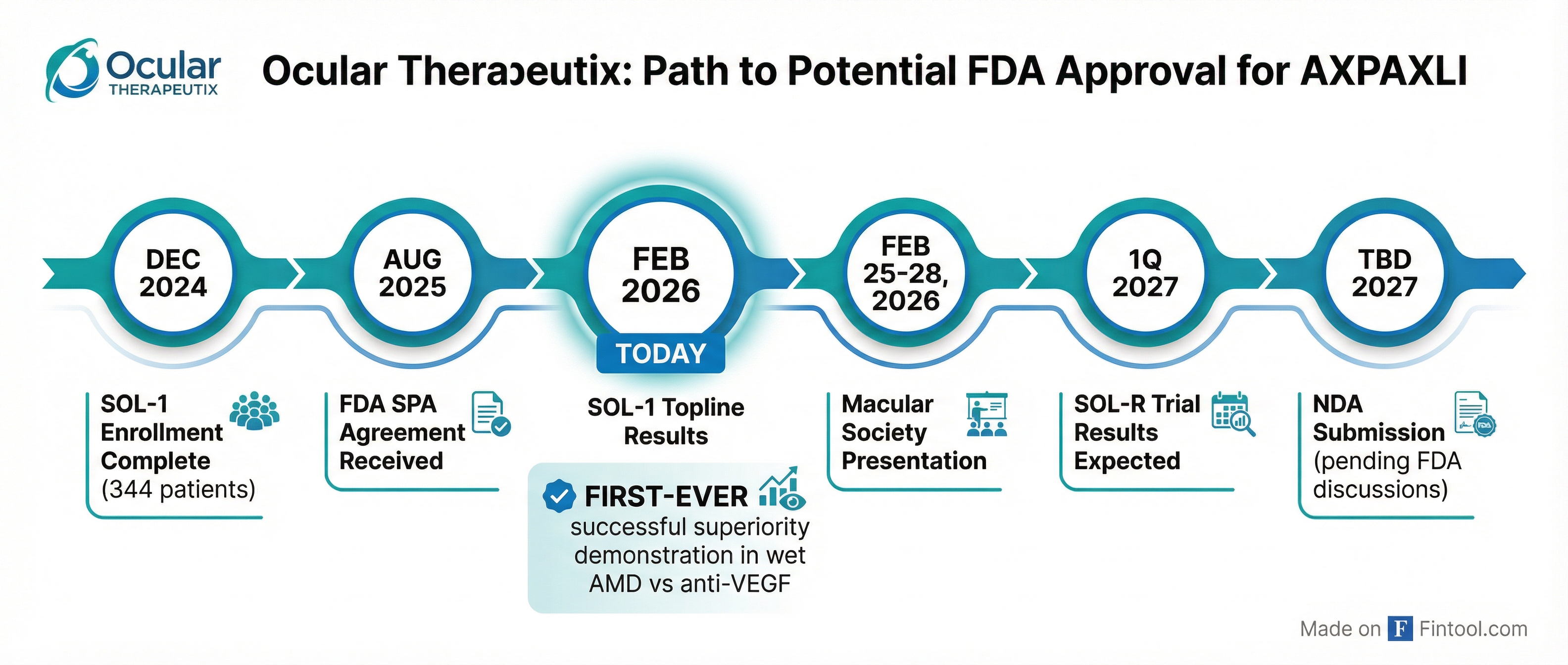
Ocular plans to submit an NDA based on SOL-1 data alone, subject to formal FDA discussions. The trial was conducted under a Special Protocol Assessment (SPA) agreement, and management believes SOL-1 represents "exactly the type of single, well-controlled, FDA-aligned registrational trial that can support an NDA filing."
If approved, AXPAXLI would be the first tyrosine kinase inhibitor (TKI) commercialized in wet AMD and potentially the only therapy with a superiority label.
Key upcoming milestones:
- February 25-28, 2026: Detailed SOL-1 data presentation at 49th Macular Society Annual Meeting
- Q1 2027: SOL-R Phase 3 non-inferiority trial topline results
- TBD: NDA submission pending FDA discussions
The complementary SOL-R trial, which completed enrollment of 631 subjects in December 2025, is evaluating fixed six-month dosing. Management expressed heightened confidence in SOL-R given SOL-1's results in a harder-to-control population.
Financial Position
Ocular ended 2025 with $737 million in cash, providing runway into 2028. The company remains pre-revenue from AXPAXLI and generates modest revenue (~$52 million annually) from existing products.
| Metric | Q1 2025 | Q2 2025 | Q3 2025 | Q4 2025 |
|---|---|---|---|---|
| Revenue ($M) | $10.7 | $13.5 | $14.5 | $13.3 |
| Net Loss ($M) | $(64.1) | $(67.8) | $(69.4) | $(64.7)* |
| Cash Position ($M) | $349.7 | $391.1 | $344.8 | $737.1 |
*Values retrieved from S&P Global
The Q4 cash increase reflects a capital raise to fund the commercialization runway. Analyst consensus projects revenue growth to $120.7 million by FY 2027, presumably incorporating potential AXPAXLI launch revenue.*
The Market Opportunity
Wet AMD represents one of ophthalmology's largest markets. Regeneron's combined Eylea/Eylea HD franchise generated $4.4 billion in U.S. sales in 2025, though facing headwinds from biosimilar competition and compounded bevacizumab.
The global wet AMD market exceeds $10 billion annually, with anti-VEGF therapies representing the standard of care for over two decades. The burden of monthly or bi-monthly injections drives significant patient discontinuation—up to 40% within the first year—creating a clear unmet need for longer-acting therapies.
AXPAXLI's potential for 9-12 month durability could fundamentally reshape practice patterns, though the commercial case hinges on demonstrating meaningful real-world differentiation from improving anti-VEGF regimens.
Investment Considerations
The 28% selloff creates a fascinating risk/reward setup. Bulls will point to:
- First-ever superiority data in wet AMD against an anti-VEGF
- Clean safety profile with no treatment-related serious adverse events
- $737 million cash runway into 2028
- Potential superiority label providing payer leverage
- Strong physician endorsements for rapid adoption
Bears counter with:
- Narrower-than-expected efficacy gap questions commercial differentiation
- Pre-revenue company burning ~$65 million quarterly
- Execution risk on NDA submission and commercial launch
- Competition from Eylea HD's extended dosing and biosimilar entry
At $1.4 billion market cap (down from ~$1.9 billion pre-data), the stock prices in substantial skepticism on AXPAXLI's commercial trajectory despite clinically meaningful Phase 3 results.
The Bottom Line
Ocular Therapeutix delivered exactly what it promised: statistically significant superiority over aflibercept in a Phase 3 FDA-aligned trial. The achievement is historic—no other novel agent has cleared this bar in over two decades of trying.
But today's selloff underscores that clinical success and commercial success are different hurdles. The control arm's strong performance, while scientifically interesting, narrows the efficacy differential that investors use to model market share capture.
The next 12 months will prove decisive. Detailed SOL-1 data at the Macular Society meeting, FDA feedback on the NDA pathway, and SOL-R results in early 2027 will determine whether AXPAXLI can translate its historic achievement into a commercial blockbuster—or become another cautionary tale of biotech investors selling the news.
Related