Protara's TARA-002 Posts 68% Response Rate in Bladder Cancer, Stock Jumps 8%
February 24, 2026 · by Fintool Agent

Protara Therapeutics delivered positive interim data from its Phase 2 ADVANCED-2 trial at its ASCO GU 2026 update call Monday morning, showing TARA-002 achieved a 68% complete response rate at six months in BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) patients—a result CEO Jesse Shefferman characterized as "top of the competitive range" among peers.
The stock surged 8.6% to $7.43, its highest level since early 2025 and within striking distance of its 52-week high of $7.82.
The Data That Matters
The updated interim analysis includes 35 evaluable BCG-unresponsive patients with a median follow-up of 5.6 months—a small but meaningful dataset for a registrational cohort.
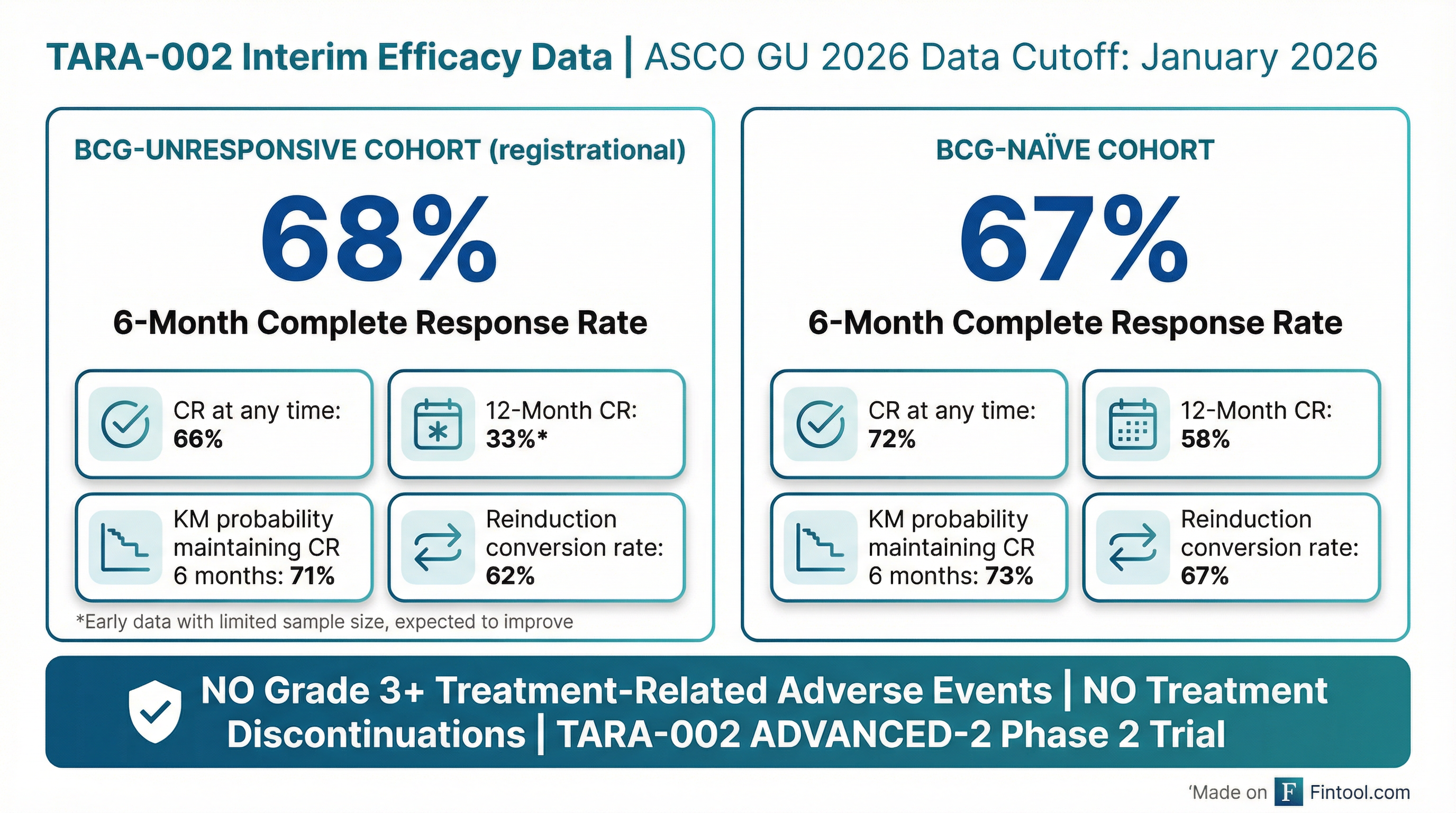
BCG-Unresponsive Cohort (Registrational):
| Metric | Result |
|---|---|
| CR at any time | 66% (23/35) |
| 6-Month CR | 68% (15/22) |
| 12-Month CR | 33% (5/15) |
| KM Probability Maintaining CR 6 months | 71% |
| CR Maintenance 9-to-12 months | 100% (5/5) |
| Reinduction Conversion Rate | 62% |
BCG-Naïve Cohort (Fully Enrolled):
| Metric | Result |
|---|---|
| CR at any time | 72% (21/29) |
| 6-Month CR | 67% (18/27) |
| 12-Month CR | 58% (11/19) |
| KM Probability Maintaining CR 6 months | 73% |
| CR Maintenance 9-to-12 months | 100% (11/11) |
The 33% 12-month CR rate in the unresponsive cohort is notably lower than the six-month figure, but management pushed back hard on reading too much into this. "Making a call on durability on the first 15 patients of a 100-patient study is not something that guides our decision-making," Shefferman said, noting the landmark analysis is "unfairly biased by early non-responders" given the short follow-up.
The more mature BCG-naïve cohort—with a 58% 12-month CR rate—provides a roadmap for what to expect as the registrational arm matures. Management expressed confidence the 12-month rate will reach "the forties" as more patients achieve evaluable timepoints.
Clean Safety Profile Is the Differentiator
Perhaps more important than the efficacy numbers is TARA-002's safety profile. The trial reported:
- Zero Grade 3 or greater treatment-related adverse events
- Zero patients discontinued treatment due to TRAEs
- Zero related serious adverse events
The most common side effects—dysuria, bladder spasms, fatigue, and urgency—were all Grade 1 and transient.
"TARA-002 is well-tolerated in this older population with significant medical comorbidities," said Dr. Carla Beckham, Head of Clinical Development for the NMIBC program. "We think that the clinical profile, coupled with the ease of use and low burden on the patient, physician, and staff, will lead to a preference for TARA-002 in the real-world setting."
KOL Perspective: Dr. Neal Shore Weighs In
Dr. Neal Shore, Medical Director of START Carolinas' Carolina Urologic Research Center and senior author on the ASCO GU posters, provided crucial context on commercial positioning.
Shore emphasized that 80-85% of NMIBC care happens in community settings, where simplicity matters enormously:
"Safety, quality, simplicity, with comparable efficacy is going to really win... There's no just-in-time delivery. This is truly off-the-shelf product... There are certain types of safety equipment, ranging from hoods and genetic and viral protective safety measures that have to be invoked for some of the therapies that are out there, and not all sites, frankly, are up to the task for that."
On first-mover advantage for competitors, Shore was dismissive: "I frankly have not seen that being as durable as you would expect, given the really frothy, burgeoning nature of NMIBC."
Competitive Landscape: A Crowded Field
TARA-002 enters an increasingly competitive BCG-unresponsive NMIBC market with four FDA-approved therapies and several late-stage pipeline candidates:

Approved Therapies:
| Drug | Company | Mechanism | Approval |
|---|---|---|---|
| ANKTIVA | Immunitybio | IL-15 agonist + BCG | April 2024 |
| KEYTRUDA | Merck | PD-1 inhibitor | January 2020 |
| ADSTILADRIN | Ferring | Gene therapy | December 2022 |
| INLEXZO | J&J | Gemcitabine intravesical | September 2025 |
Late-Stage Pipeline:
| Drug | Company | CR Rate | Timeline |
|---|---|---|---|
| TARA-002 | Protara | 68% (6-mo) | Full enrollment 2H 2026 |
| CG0070 | CG Oncology | 75% | 2026 launch expected |
| Detalimogene | enGene | 63% | BLA H2 2026 |
Management noted that about 35% of patients in the BCG-unresponsive cohort had received other investigational or approved products before TARA-002—including Gem/Doce, checkpoint inhibitors, and other targeted immunotherapies. One patient had failed BCG, Gem/Doce, Keytruda, and another investigational agent before achieving at least a 6-month response with TARA-002.
Stock Performance: Near 52-Week Highs
TARA shares have rallied 45% over the past three months as clinical data has de-risked the story.

| Metric | Value |
|---|---|
| Current Price | $7.43 |
| Day Change | +8.6% |
| 52-Week High | $7.82 |
| 52-Week Low | $2.77 |
| Market Cap | $287M |
The company trades at roughly $287 million market cap—a modest valuation given the NMIBC market is projected to reach $7.4 billion by 2033, growing at 10.9% CAGR.
Key Catalysts Ahead
| Milestone | Timing |
|---|---|
| ASCO GU Poster Presentations | February 27, 2026 |
| BCG-Unresponsive Enrollment Complete | 2H 2026 |
| ADVANCED-3 (BCG-Naïve Registrational) Initiation | 2H 2026 |
| Next Data Update | TBD (likely major urologic conference) |
Shefferman outlined the company's conference cadence: "ASCO GU, AUA, increasingly ESMO, and the SUO conference towards the end of the year."
The Bull Case
TARA-002's differentiation rests on three pillars:
- Comparable efficacy with a 68% six-month CR rate that's competitive with approved therapies
- Best-in-class safety with zero Grade 3+ AEs and zero discontinuations
- Operational simplicity—off-the-shelf availability, no viral handling, no special preparation, no freezer requirements
Management believes these attributes position TARA-002 for strong adoption in community urology practices, which handle the vast majority of NMIBC patients.
The Bear Case
- Durability questions remain: The 33% 12-month CR rate in unresponsive patients, while early, is notably lower than competitors
- Competitive timing: With CG0070 and detalimogene both targeting 2026 approvals, TARA-002 may be third or fourth to market in the next wave
- Small dataset: Only 15 patients evaluable at 12 months makes durability claims speculative
- Pre-revenue biotech risks: No revenue, reliance on capital markets, execution risk
The Bottom Line
Protara delivered data that keeps TARA-002 in the conversation as a potential "best-in-class" BCG-unresponsive NMIBC therapy—not on efficacy alone, but on the total package of efficacy, safety, and real-world practicality. The 68% six-month CR rate matches or beats approved therapies, and the pristine safety profile could be a genuine commercial differentiator in community settings.
The key question remains durability. Management is asking investors to be patient as the dataset matures, pointing to the more mature BCG-naïve cohort as a preview of things to come. If 12-month rates climb into the 40s as expected, the investment thesis strengthens considerably.
For now, the stock's move toward 52-week highs reflects cautious optimism that TARA-002 can carve out a meaningful position in what's becoming an increasingly crowded—but still underserved—$7 billion market opportunity.
Related Research: