Vir Biotechnology Surges 55% on $1.7 Billion Astellas Deal for Prostate Cancer T-Cell Engager
February 23, 2026 · by Fintool Agent
Vir Biotechnology stock surged 55% in after-hours trading after the company announced a global strategic collaboration with Astellas Pharma worth up to $1.7 billion for VIR-5500, a dual-masked T-cell engager targeting prostate cancer that posted compelling Phase 1 data with an 82% PSA50 response rate and 45% objective response rate at the highest doses tested.
The deal marks a seminal moment for Vir as it validates the company's strategic pivot toward oncology under CEO Marianne De Backer, who licensed the PRO-XTEN masking technology platform from Sanofi in 2024 to build an immunotherapy pipeline in solid tumors.
The Deal Economics
Vir will receive $335 million in upfront and near-term payments, structured as $240 million in cash, $75 million in equity investment at a 50% premium to the 30-day volume-weighted average price, and a $20 million milestone upon completion of manufacturing technology transfer expected in Q2-Q3 2027.
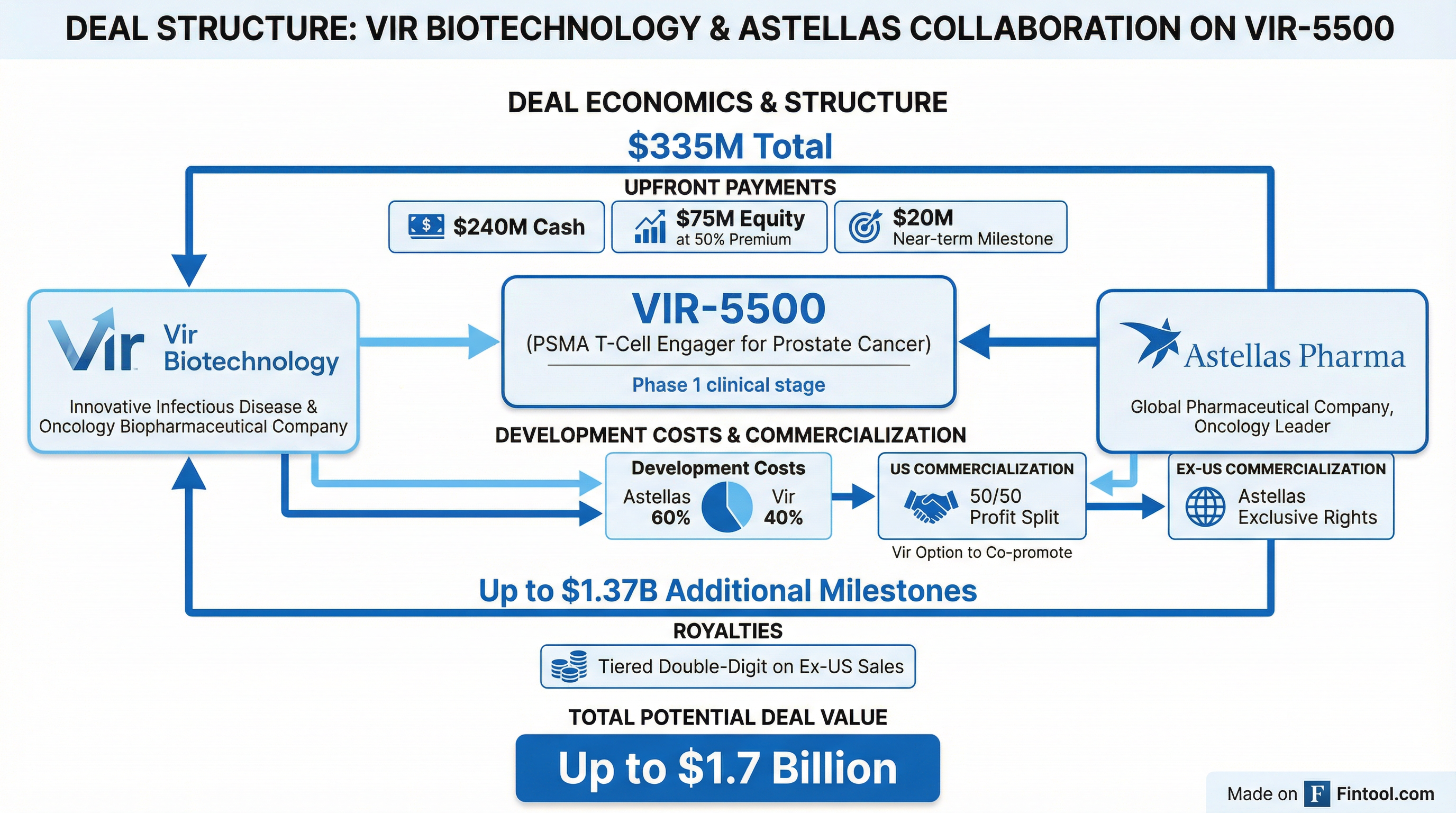
Beyond the upfront payment, Vir is eligible for up to $1.37 billion in additional development, regulatory, and sales milestones, along with tiered double-digit royalties on ex-US net sales.
| Component | Value |
|---|---|
| Upfront Cash | $240M |
| Equity Investment (50% premium) | $75M |
| Near-Term Milestone | $20M |
| Total Upfront | $335M |
| Additional Milestones | Up to $1.37B |
| Ex-US Royalties | Tiered double-digit |
| Total Deal Value | Up to $1.7B |
Under the agreement, global development costs will be shared with Astellas responsible for 60% and Vir for 40%. In the US, Vir retains the option to co-promote and will share profits and losses equally with Astellas. Outside the US, Astellas obtains exclusive commercialization rights.
Phase 1 Data: Best-in-Class Potential
The collaboration announcement coincided with updated Phase 1 data that will be presented at the 2026 ASCO Genitourinary Cancers Symposium on February 26. The results support VIR-5500's potential as a best-in-class PSMA-targeting T-cell engager.
Across 58 patients receiving VIR-5500 monotherapy, the drug was generally well tolerated with no dose-limiting toxicities observed. Grade 3 or higher treatment-related adverse events occurred in just 12% (7/58) of patients and were manageable.
Key Efficacy Data (≥3,000 μg/kg Q3W dosing cohorts, n=22):
| Endpoint | Result |
|---|---|
| PSA50 Response (≥50% PSA decline) | 82% (14/17 evaluable) |
| PSA90 Response (≥90% PSA decline) | 53% (9/17 evaluable) |
| Objective Response Rate (RECIST) | 45% (5/11 evaluable) |
| Confirmed Responses | 4 of 5 responders |
Critically, cytokine release syndrome (CRS)—a common and potentially dangerous side effect of T-cell engagers—was limited, occurring in 50% of patients but generally restricted to Grade 1 (fever only). Prophylactic steroids were not required and no IL-6 inhibitors were used to manage CRS.
"It is remarkable to see these early signs of profound anti-tumor activity in heavily pre-treated mCRPC patients, and the favorable tolerability with minimal CRS to date means VIR-5500 could play a role in treating earlier disease," said Dr. Johann de Bono, Principal Investigator and Director of the Drug Development Unit at the Institute of Cancer Research.
Why This Matters: The Prostate Cancer Opportunity
Prostate cancer remains the most common cancer diagnosis in men and the second leading cause of cancer-related mortality in men behind lung cancer. Metastatic castration-resistant prostate cancer (mCRPC)—where patients have progressed despite hormonal therapies—has a 5-year survival rate of approximately 30%.
For Astellas, this deal reinforces its prostate cancer leadership as its blockbuster Xtandi (enzalutamide) begins to face patent expiration. Xtandi, partnered with Pfizer, has treated over 1.5 million patients and generated approximately $6 billion in revenue.
"Astellas is proud to have helped 1.5 million patients with prostate cancer, and we are dedicated to expanding our impact as part of our R&D strategy," said Adam Pearson, Chief Strategy Officer at Astellas. "This strategic collaboration allows Astellas and Vir Biotechnology to combine our expertise and reaffirms our commitment to improving the lives of people with prostate cancer."
Competitive Landscape: Vir vs. Janux
Janux Therapeutics is developing a competing PSMA-targeting T-cell engager (JANX007) with single-masking technology. In December, Janux reported Phase 1 data showing PSA50 of 86% and objective response rate of 30% among evaluable patients at its selected dosing regimen.
| Metric | VIR-5500 | JANX007 |
|---|---|---|
| PSA50 Response | 82% | 86% |
| Objective Response Rate | 45% | 30% |
| Masking Technology | Dual-masked (PRO-XTEN) | Single-masked |
| Prophylactic Steroids Required | No | Not disclosed |
Vir's dual-masking approach—which covers both the tumor antigen side and the CD3 component—may offer additional protection before reaching the target tumor, potentially enabling a better therapeutic index.
Stock Reaction and Runway Extension
VIR shares closed at $7.43 on February 23 before the after-hours announcement. The 55% surge values the equity investment at a significant premium to the prior trading price.
Importantly, the deal extends Vir's cash runway significantly. The company reported $781.6 million in cash and investments as of December 31, 2025, and now expects funding to last into the second quarter of 2028 including the expected net effects of the Astellas collaboration.
| Financial Metric | FY 2024 | FY 2023 |
|---|---|---|
| Revenues | $74.2M | $86.2M |
| Net Loss | $(522.0M) | $(615.1M) |
| Cash & Equivalents | $222.9M | $241.6M |
For full year 2025, Vir reported a net loss of $438.0 million, a significant improvement from $(521.96) million in 2024, driven by cost savings from restructuring initiatives.
Development Timeline and What to Watch
Vir has concluded monotherapy dose-escalation and defined a preliminary go-forward dose. The company plans to:
- Q2 2026: Initiate monotherapy dose-expansion cohorts in late-line mCRPC
- Q2 2026: Initiate combination dose-expansion cohorts in early-line mCRPC and mHSPC
- February 26, 2026: Present oral abstract at ASCO GU Symposium
- 2027: Initiate pivotal Phase 3 trials
The dose-escalation of VIR-5500 in combination with enzalutamide continues in early-line mCRPC patients, potentially expanding the addressable market beyond late-stage disease.
The Bottom Line
The Astellas deal represents a transformative moment for Vir Biotechnology, validating its oncology pivot and providing substantial non-dilutive capital to advance its pipeline. The Phase 1 data for VIR-5500 show a potentially differentiated safety and efficacy profile compared to competing T-cell engagers, particularly on the tolerability front where the lack of required prophylactic steroids could be a meaningful clinical advantage.
For Astellas, the deal fills a critical gap in its prostate cancer franchise as Xtandi faces patent erosion. The 60/40 development cost split and US profit-sharing structure demonstrate confidence in VIR-5500's commercial potential while managing risk.
The key question now is whether the Phase 1 efficacy signals translate to pivotal studies. With Phase 3 trials expected in 2027, investors will be watching the ASCO GU presentation on February 26 and subsequent expansion cohort data closely.
Related: