AbbVie Doubles Down on U.S. Manufacturing with $380 Million API Investment
February 23, 2026 · by Fintool Agent

Abbvie is pouring $380 million into two new active pharmaceutical ingredient manufacturing facilities at its North Chicago headquarters, accelerating the pharma giant's push to bring drug production back to American soil—and positioning itself to manufacture next-generation obesity medicines domestically.
The investment marks another milestone in Abbvie's $100 billion pledge to U.S.-based R&D and capital investments over the next decade—a commitment made in January under a voluntary agreement with the Trump administration that secured the company a three-year exemption from tariffs and future price mandates.
ABBV shares rose 2.1% Monday to $229.48, adding $8 billion in market cap.
The Investment
Construction begins this spring, with both facilities expected to be fully operational by 2029.
| Detail | Specification |
|---|---|
| Investment | $380 million |
| Facilities | Two state-of-the-art API plants |
| Location | North Chicago, Illinois campus |
| Jobs Created | 300 (engineers, scientists, operators, technicians) |
| Construction Start | Spring 2026 |
| Operational Target | 2029 |
| Focus | Neuroscience and obesity medications |
The new facilities will integrate advanced manufacturing technologies with artificial intelligence to support production of AbbVie's pipeline of neuroscience and obesity drugs—two therapeutic areas CEO Rob Michael has emphasized as core growth drivers for the next decade.
"This milestone demonstrates further progress against our $100 billion commitment to U.S. R&D and capital investments over the next decade," said Robert A. Michael, chairman and CEO. "By strengthening our U.S. manufacturing capabilities, we are well-positioned to support our investment in innovation and enhance our ability to deliver next-generation medicines to patients."
The $100 Billion Deal
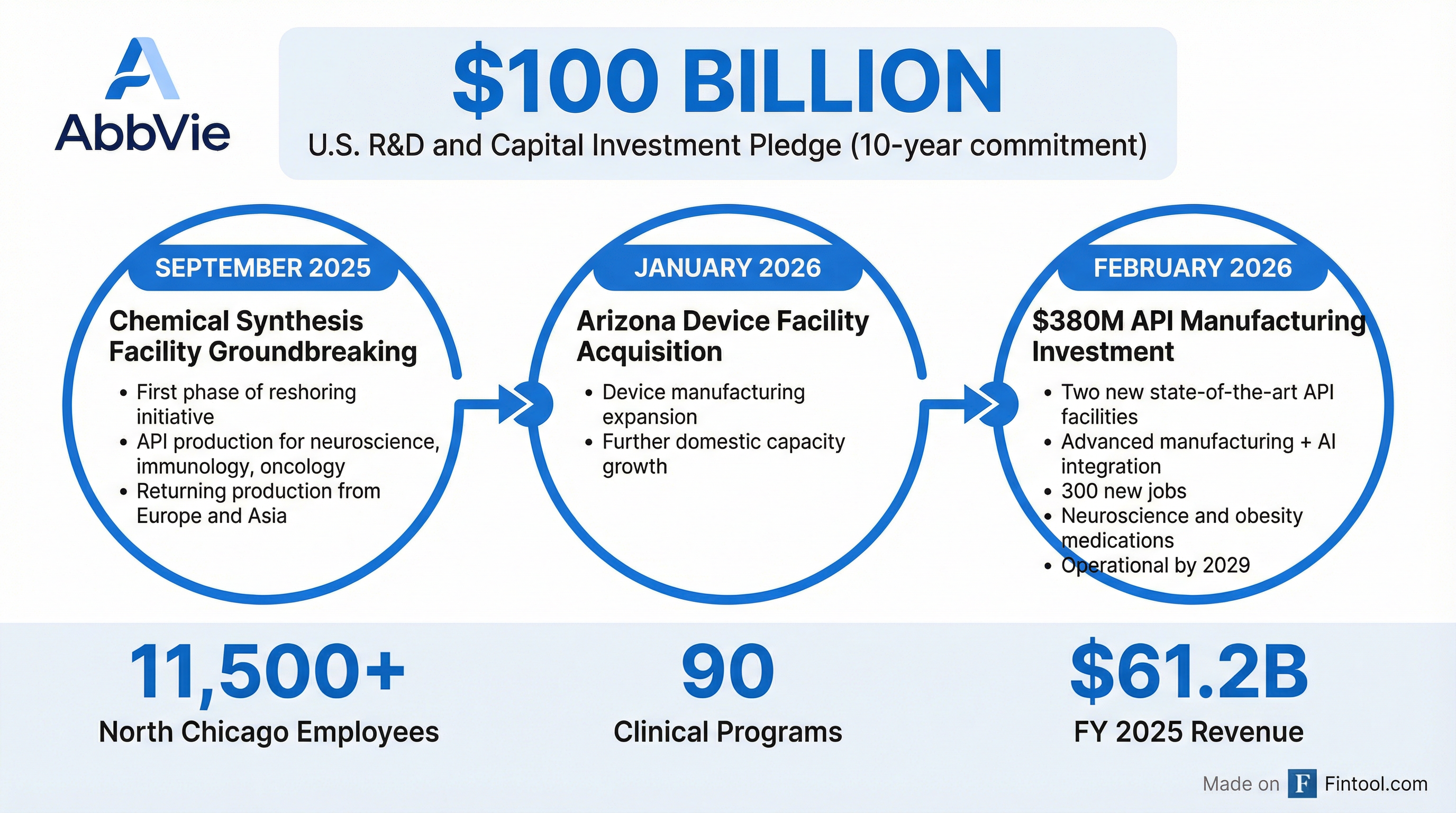
Today's announcement continues momentum from a reshoring initiative that began in September 2025, when AbbVie broke ground on its first chemical synthesis facility—designed to bring API production for select neuroscience, immunology, and oncology products back from Europe and Asia. In January 2026, the company announced the acquisition of a device manufacturing facility in Arizona.
The voluntary agreement with the U.S. government includes significant concessions from both sides. AbbVie committed to:
- $100 billion in U.S.-based R&D and capital investment over ten years
- Low prices in Medicaid programs
- Expanded affordable, direct-to-patient offerings
In return, the government agreed to provide AbbVie a three-year exemption from tariffs and future price mandates—a significant shield as the company navigates increasing pricing pressure from the Inflation Reduction Act, which has already selected Imbruvica, Vraylar, Linzess, and Botox for Medicare price negotiations.
Illinois Governor JB Pritzker praised the expansion, announcing it in conjunction with the Illinois Department of Commerce and Economic Opportunity.
Why Obesity and Neuroscience?
AbbVie is making a strategic bet on obesity—a therapeutic area it entered through business development in 2025 with the acquisition of ABBV-295, a long-acting amylin analog. The facilities announced today will be purpose-built to manufacture APIs for these next-generation treatments.
"We're clearly taking a close look at what's available out there," CEO Michael said on the recent earnings call regarding obesity assets. "To the extent we see a differentiated asset in any of our core areas, whether early or late stage... we are willing to invest. We certainly have the financial wherewithal."
The company's pipeline now includes approximately 90 compounds, devices, or indications in development, with roughly 60 in mid- and late-stage trials. Core therapeutic focus areas include:
- Immunology (Skyrizi, Rinvoq—combined sales guidance >$31 billion in 2026)
- Neuroscience (next-gen assets 932, Bretisilocin, Emraclidine)
- Oncology (Etentamig registrational data expected)
- Obesity (ABBV-295 and potential future acquisitions)
Financial Position
AbbVie enters this capital-intensive expansion phase from a position of strength:
| Metric | Q1 2025 | Q2 2025 | Q3 2025 | Q4 2025 |
|---|---|---|---|---|
| Revenue ($B) | $13.3 | $15.4 | $15.8 | $16.6 |
| Net Income ($B) | $1.3 | $0.9 | $0.2 | $1.8 |
| EBITDA Margin % | 45.7% | 50.8% | 48.6% | 46.1% |
| CapEx ($M) | -$235 | -$269 | -$381 | -$329 |
Full-year 2025 delivered record net revenues of $61.2 billion—up 9%—and operating cash flows of $19 billion. The company invested $1.2 billion in capital expenditures during the year and over $5 billion in external business development.
Management has guided for 9.5% revenue growth in 2026, driven by Skyrizi and Rinvoq exceeding $31 billion in combined sales—already $500 million ahead of the 2027 long-term guidance issued just a year ago.
The Bigger Picture: Pharma Reshoring
AbbVie's manufacturing push reflects a broader industry trend as pharmaceutical companies respond to both policy incentives and supply chain lessons from recent years. The 2025 Act, signed into law on July 4, 2025, restored favorable tax treatment for domestic R&D costs and bonus depreciation—making U.S. manufacturing more attractive.
The company noted it is "currently in discussions with multiple U.S. states about potential manufacturing investments and anticipates announcing additional investments in 2026."
With more than 11,500 employees at its North Chicago headquarters and approximately 29,000 across the U.S., AbbVie is already one of the largest pharmaceutical employers in the country. These new facilities will add 300 more.
What to Watch
Near-term catalysts:
- Spring 2026 construction commencement
- Additional state-level manufacturing announcements expected
- Pipeline readouts for neuroscience assets (932, Bretisilocin, Emraclidine) in next 24 months
- Obesity pipeline expansion through potential M&A
Investor considerations:
- How quickly can AbbVie ramp its obesity portfolio to compete with Eli Lilly and Novo Nordisk?
- Will the three-year tariff exemption prove sufficient as trade tensions escalate?
- Can the $100 billion commitment drive competitive advantage in domestic supply chains?
Related Companies: Abbvie | Eli Lilly | Novo Nordisk