MoonLake's Investor Day Delivers 81% Response Rate in Spine Disease—Best Ever in Clinical Trial
February 23, 2026 · by Fintool Agent

Moonlake Immunotherapeutics unveiled what management called "diamond" Phase 2 data at its Investor Day today, reporting that 81% of patients in its S-OLARIS trial achieved ASAS40 response at week 12—the highest 12-week response rate ever reported in an axial spondyloarthritis (axSpA) clinical trial.
The data, presented alongside regulatory clarity on the company's hidradenitis suppurativa (HS) approval path, sent shares up as much as 10% in premarket trading before settling lower on the session.
For a stock that collapsed 90% in a single day last September, today's event marks a potential inflection point—and signals that MoonLake's nanobody platform may deliver on its promise across multiple large inflammatory disease markets.
The Data: "Small But Beautiful"
Chief Scientific Officer Kristian Reich called the S-OLARIS study a "diamond" that "really shines" with multiple validating endpoints beyond traditional clinical scores.
The trial's headline numbers were striking:
| Endpoint | Week 12 Result | Competitor Reference |
|---|---|---|
| ASAS40 Response | 81% | 50% (existing IL-17s) |
| ASDAS-CRP (low disease) | 80%+ | 60% (long-term competitor data) |
| SPARCC MRI Reduction | >80% | -8 to -10 absolute (best-in-class) |
| PET Osteoblast Activity | >40% reduction | First drug with PET validation |
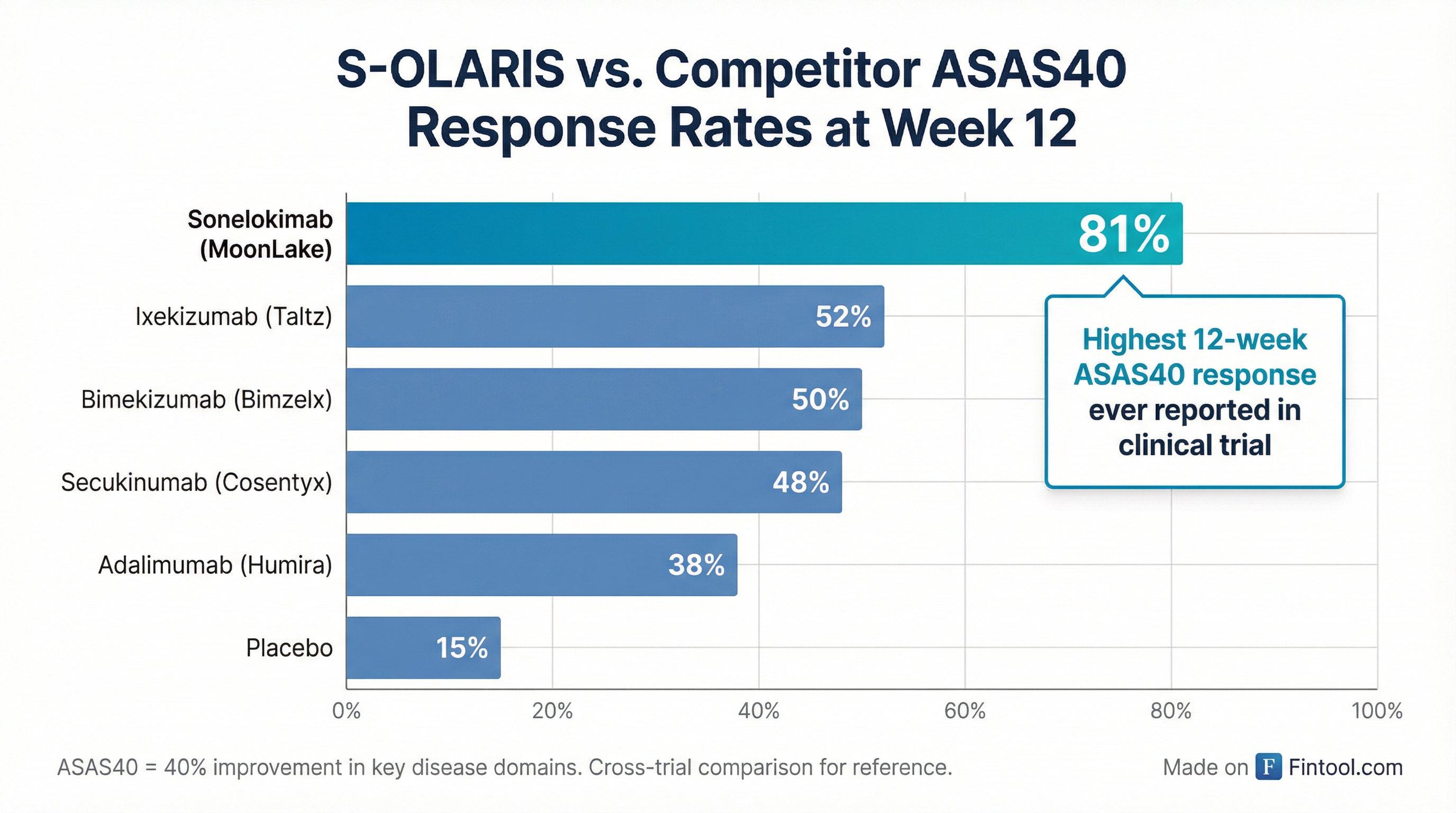
What makes the data particularly compelling is the multi-modal validation. While competitor trials typically rely on patient-reported questionnaires (which carry placebo response risk), MoonLake designed S-OLARIS with MRI, PET imaging, and biomarker programs to objectively verify clinical improvements.
"You may see a placebo response in a patient questionnaire. You will not see a placebo response in structural damage, PET imaging, or these biomarkers," Reich explained.
The PET imaging specifically measured osteoblast activity—the biological driver of the irreversible bone fusion that defines axSpA's worst outcomes. A 40%+ reduction suggests sonelokimab may modify disease progression, not just symptoms.
The HS Path: Narrow But Clear
Perhaps more important for near-term valuation was CEO Jorge Santos da Silva's detailed walkthrough of the company's FDA regulatory path for hidradenitis suppurativa—MoonLake's lead indication.
The company received Type B meeting guidance from the FDA in December/January that management described as creating a "very narrow, very clear path" to approval:
- VELA-1 and MIRA establish substantial evidence of effectiveness (SEE)
- VELA-2 supports safety, with possible inclusion in efficacy TBD
- No additional trials required
- BLA submission targeted for end of Q3 2026
This matters because MoonLake's September 2025 crash stemmed from VELA-2 missing its primary endpoint—a delta to placebo that fell short of statistical significance despite strong absolute response rates. The FDA guidance essentially confirms the regulatory path remains intact.
Santos da Silva walked through potential label scenarios, suggesting MoonLake's efficacy numbers would compare favorably to UCB's bimekizumab even in a base case:
| Drug | HiSCR75 Response | HiSCR50 Response |
|---|---|---|
| Sonelokimab (VELA-1) | 45% | 58% |
| Sonelokimab (MIRA) | 47% | 59% |
| Bimekizumab (BE HEARD) | 30-35% | 45-48% |
"We believe Section 14 of the label we're going to propose can be really a leading profile versus many of our competitors," Santos da Silva stated.
The Recovery Trade
MoonLake's stock tells a remarkable turnaround story. After trading above $60 in late September 2025, shares crashed to $6.25 on the VELA-2 miss—a 90% single-day collapse that wiped out years of gains.
The stock has since recovered 188% from those lows, driven by:
- January 8, 2026: FDA Type B meeting confirms regulatory path
- February 2, 2026: FDA grants Fast Track designation for PPP
- February 22, 2026: S-OLARIS Phase 2 data release
With 12.7% of float sold short, the setup creates potential for further squeeze if positive catalysts continue.
The Financial Picture
CFO Matthias Bodenstedt reported the company ended Q4 2025 with $394 million in cash, cash equivalents, and short-term marketable securities—runway extending into H2 2027.
| Metric | Q4 2025 | Q3 2025 |
|---|---|---|
| Cash Position | $394M | — |
| R&D Expense | $56.0M | $60.6M |
| G&A Expense | $9.2M | $10.8M |
| Total OpEx | $65.2M | $71.4M |
The company also amended its Hercules Capital debt facility, drawing $25 million and retaining access to $400 million in additional non-dilutive funding for commercial preparation.
"That will not only help us with the future development of sonidegib, but will then therefore and primarily also contribute to the commercial readiness," Bodenstedt explained.
What's Next: A Catalyst-Rich Calendar
MoonLake faces a packed 2026 with multiple binary events:
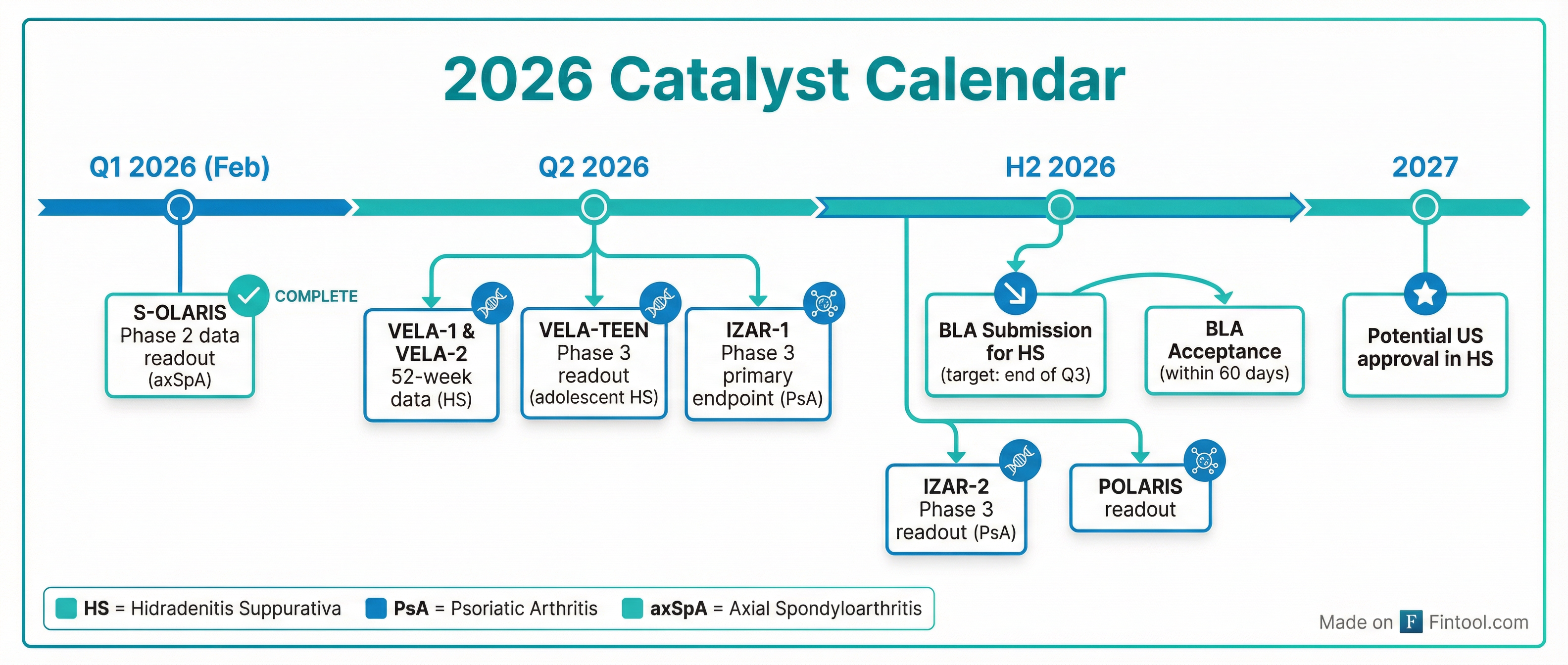
Q2 2026:
- VELA-1 and VELA-2 52-week long-term data (HS)
- VELA-TEEN Phase 3 primary endpoint (adolescent HS)
- IZAR-1 Phase 3 primary endpoint (psoriatic arthritis)
H2 2026:
- BLA submission for HS (end of Q3)
- BLA acceptance (within 60 days)
- IZAR-2 Phase 3 readout (PsA)
The IZAR-1 readout in PsA will be particularly watched. The Phase 2 ARGO data showed sonelokimab achieved high ACR70 + PASI100 combination responses—a bar few competitors reach.
Competitive Positioning
MoonLake's main competitor in IL-17A/F inhibition is UCB's bimekizumab (Bimzelx), which received FDA approval for HS in November 2024.
MoonLake argues its nanobody technology—a 40kDa construct versus traditional ~150kDa antibodies—provides several advantages:
- Deep tissue penetration: Smaller size enables access to inflammatory sites
- Albumin binding: Extended half-life and enrichment at inflammation sites
- Convenient dosing: 1mL monthly maintenance versus more frequent competitor regimens
Management highlighted potential safety differentiation as well, noting no signals for liver enzyme elevations, IBD, or suicidal ideation that appear in bimekizumab's label.
"We're always injecting far less number of molecules into every patient to produce a similar or better result," Santos da Silva noted.
The Bottom Line
MoonLake's Investor Day accomplished what it needed to: validate the platform beyond HS, clarify the regulatory path, and demonstrate that the September collapse may have been an overreaction.
The S-OLARIS data, if replicated in Phase 3, could position sonelokimab as a disease-modifying therapy in axSpA—a category distinction that would differentiate it from symptom-focused competitors. The company's CSO captured the ambition: "This is how I would see the axSpA data. Small but beautiful."
For investors, the risk/reward has shifted. With regulatory clarity, strong efficacy data, and a funded runway, MoonLake's path to commercialization is narrower—but arguably clearer—than it was six months ago.
The next major test comes in Q2 with IZAR-1 in psoriatic arthritis. A second Phase 3 win would cement the nanobody thesis and potentially unlock the $10-15 billion axSpA market management believes is within reach.
Related
- Moonlake Immunotherapeutics
- Novartis (Cosentyx/secukinumab)
- Abbvie (Humira/adalimumab)
- Johnson & Johnson (Tremfya)