Lilly Proves Zepbound Boosts Psoriasis Treatment: GLP-1s Expand Beyond Weight Loss
February 19, 2026 · by Fintool Agent

Eli Lilly just delivered clinical proof that GLP-1 drugs can do more than shrink waistlines—they can make other treatments work better.
In the Phase 3b TOGETHER-PsO trial, patients taking Lilly's Zepbound alongside its psoriasis drug Taltz achieved a 40% relative improvement in complete skin clearance compared to Taltz alone (40.6% vs. 29.0%), meeting the study's key secondary endpoint. The primary endpoint was even more striking: 27.1% of combination therapy patients achieved both complete skin clearance and at least 10% weight loss, compared to just 5.8% on Taltz monotherapy.
This is the second positive readout from Lilly's TOGETHER program in two months, following January's psoriatic arthritis data that showed a 64% relative improvement in disease activity with the combination. Together, these results make Taltz the first and only biologic with clinical evidence supporting concomitant use with a GLP-1 for patients who have both psoriatic disease and obesity.
"These PASI 100 results represent far more than a clinical milestone—they demonstrate what becomes possible when we address both simultaneously," said Adrienne Brown, Lilly's executive vice president and president of immunology.
The Trial Results
The TOGETHER-PsO study enrolled 274 adults with moderate-to-severe plaque psoriasis who also had obesity (BMI ≥30) or overweight (BMI 27-29.9) with at least one weight-related comorbidity. Participants were randomized 1:1 to receive either Taltz alone or Taltz plus Zepbound, with both groups receiving diet and exercise counseling.
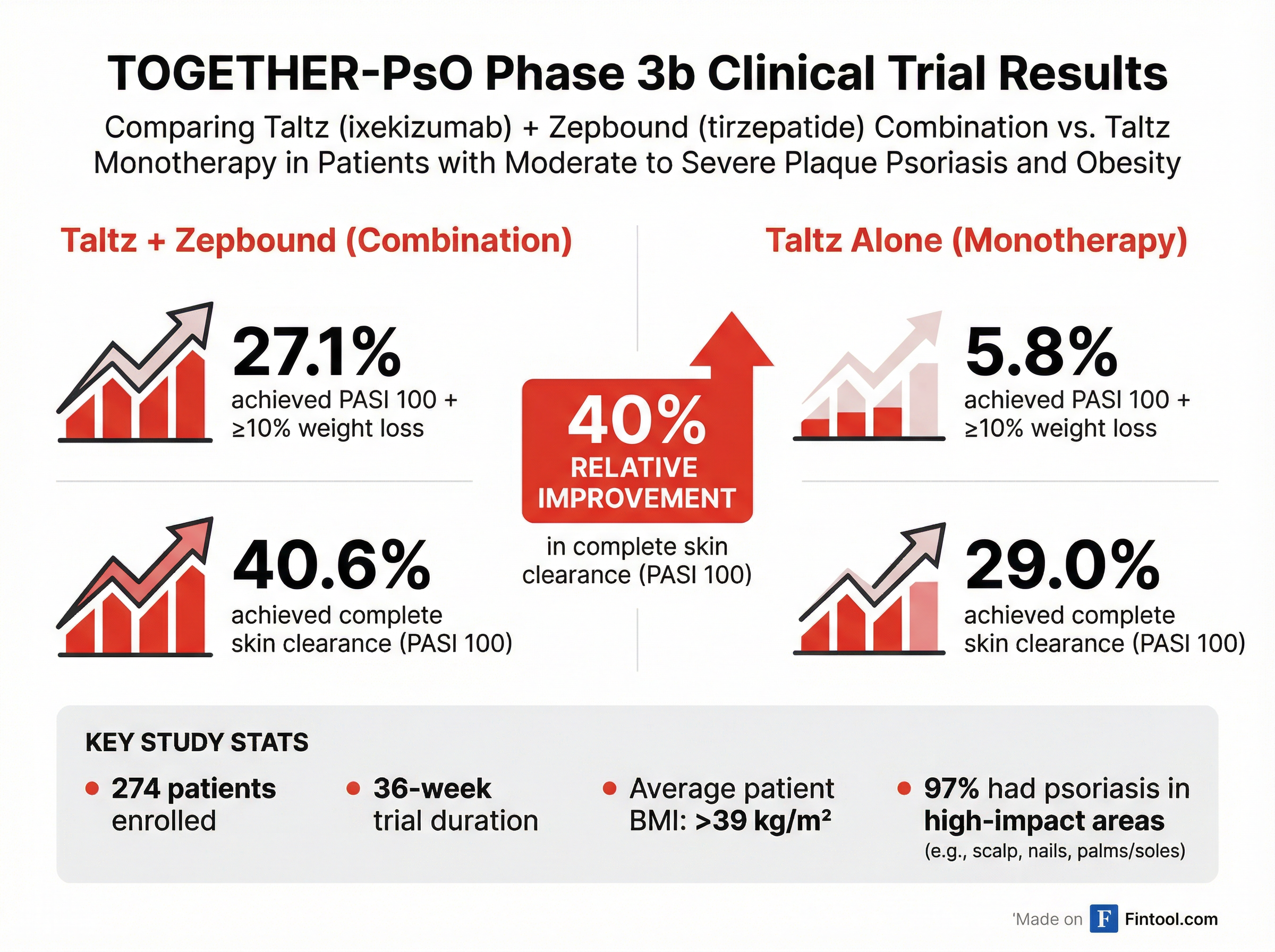
The study population had a particularly high disease burden. The average BMI exceeded 39 kg/m²—approximately 9-10 kg/m² higher than any population studied in Phase 3 pivotal trials of a psoriasis biologic. Nearly all participants (97%) had psoriasis affecting high-impact body areas like the face, scalp, or genitals, and approximately 25% of their body surface area was affected.
| Endpoint | Taltz + Zepbound | Taltz Alone | Relative Improvement |
|---|---|---|---|
| PASI 100 + ≥10% weight loss (Primary) | 27.1% | 5.8% | 367% (p<0.001) |
| Complete skin clearance (PASI 100) | 40.6% | 29.0% | 40% (p<0.05) |
"This study involved patients with particularly high BMI and difficult-to-treat psoriasis, making the PASI 100 results with Taltz plus Zepbound especially remarkable," said Dr. Mark Lebwohl, the study's principal investigator and Dean for Clinical Therapeutics at Mount Sinai's Icahn School of Medicine.
Why This Matters: GLP-1s as Inflammation Fighters
The clinical significance extends beyond the headline numbers. The data suggests that treating obesity with Zepbound doesn't just help patients lose weight—it directly reduces the burden of psoriatic disease through both weight-dependent and weight-independent mechanisms.
"Psoriasis and obesity share underlying inflammatory pathways, yet they are too often treated in silos despite psoriasis treatment guidelines calling for obesity management," Dr. Lebwohl noted.
This represents a potential paradigm shift. Approximately 61% of U.S. psoriasis patients also have obesity or overweight with at least one weight-related comorbidity. For psoriatic arthritis, that figure is 65%. Higher BMI has been shown to reduce the odds of reaching skin clearance across multiple psoriasis studies, making these patients historically harder to treat.
The January TOGETHER-PsA results in psoriatic arthritis were similarly compelling:
| TOGETHER-PsA Results (Jan 2026) | Taltz + Zepbound | Taltz Alone |
|---|---|---|
| ACR50 + ≥10% weight loss (Primary) | 31.7% | 0.8% |
| ACR50 (50% improvement in PsA activity) | 33.5% | 20.4% |
| Relative improvement in ACR50 | — | 64% |
"An important finding from the study was the potential effect of tirzepatide when added to ixekizumab on psoriatic arthritis symptoms via both weight-dependent and also weight-independent mechanisms," said Dr. Dan Skovronsky, Lilly's Chief Scientific Officer, on the Q4 2025 earnings call.
Lilly's Broader Incretin + Immunology Strategy
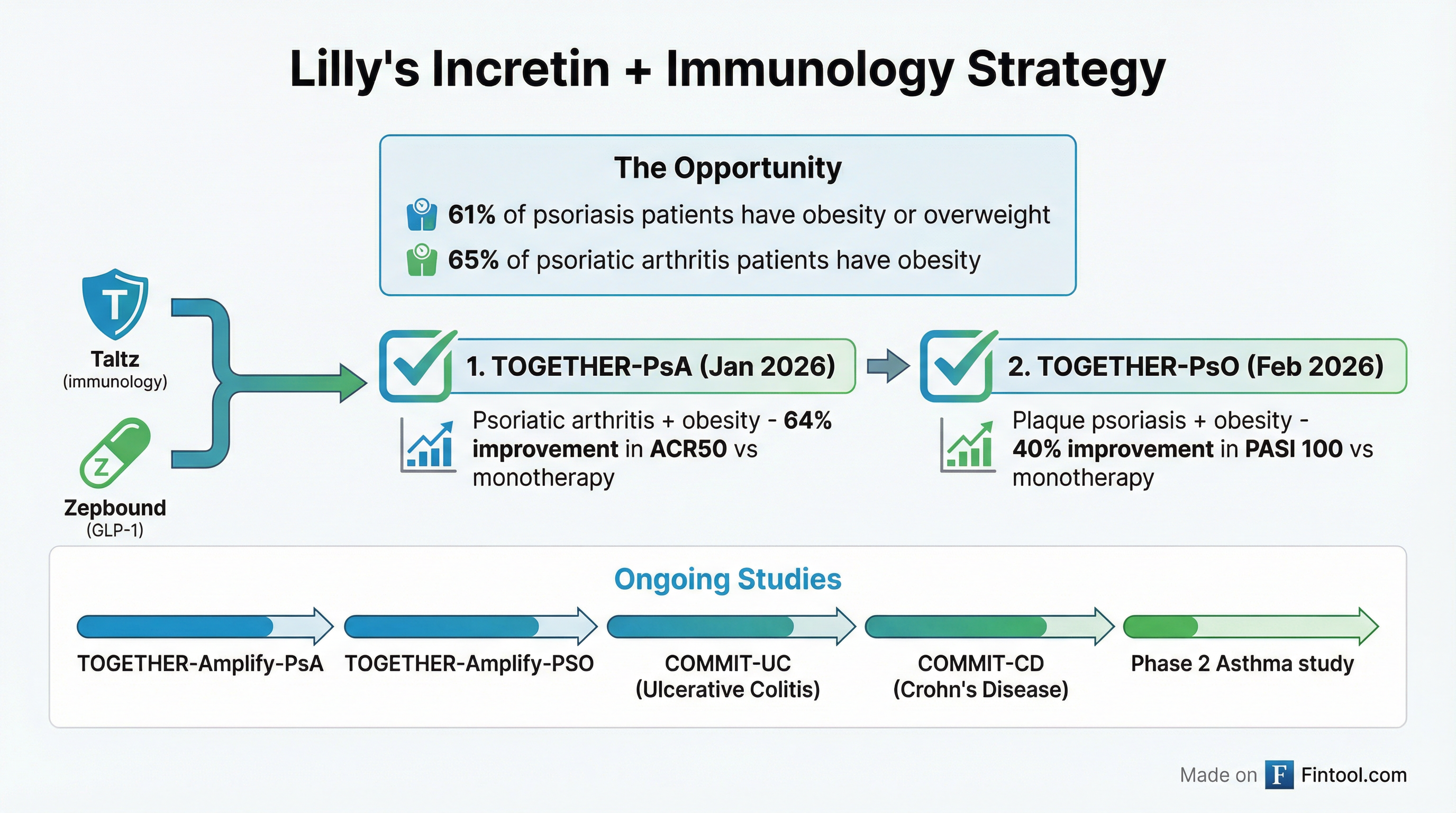
The TOGETHER trials are part of a broader strategic push by Lilly to leverage its dominance in incretins (Zepbound, Mounjaro) to unlock better outcomes across immunology. The company is running an extensive pipeline of combination studies:
Completed:
- TOGETHER-PsA (psoriatic arthritis) — Positive Jan 2026
- TOGETHER-PsO (plaque psoriasis) — Positive Feb 2026
Ongoing:
- TOGETHER-Amplify-PsA (adding Zepbound after starting Taltz in psoriatic arthritis)
- TOGETHER-Amplify-PSO (adding Zepbound after starting Taltz in psoriasis)
- COMMIT-UC (Omvoh + Zepbound in ulcerative colitis)
- COMMIT-CD (Omvoh + Zepbound in Crohn's disease)
- Phase 2 study of tirzepatide in asthma
"More than a billion people worldwide have immune diseases like atopic dermatitis, psoriasis, IBD, and asthma. But patients who have both immune diseases and obesity tend to have a higher disease burden," Adrienne Brown explained on the earnings call. "We're really excited about the opportunity to find new ways to combat the underlying inflammation in these diseases."
Financial Context
Lilly enters these new therapeutic opportunities from a position of strength. Q4 2025 revenue hit $19.3 billion, with full-year growth driven by the Zepbound/Mounjaro franchise.
| Metric | Q4 2025 | Q3 2025 | Q2 2025 | Q1 2025 |
|---|---|---|---|---|
| Revenue | $19.3B | $17.6B | $15.6B | $12.7B |
| Gross Margin | 82.5% | 82.9% | 84.3% | 82.5% |
The stock barely moved on the announcement—LLY closed at $1,023.22 on February 19, up 0.26%, after falling 1.14% on the announcement day. The muted reaction likely reflects the market having already anticipated positive results following January's TOGETHER-PsA success.
At a market cap of roughly $965 billion, Lilly trades at approximately 50x forward earnings, a premium valuation that already incorporates expectations for the GLP-1 franchise to expand beyond obesity and diabetes.
Safety Profile
Adverse events in the combination arm were generally mild to moderate and consistent with the known safety profiles of both drugs. The most common side effects (≥5% of participants) in the Taltz plus Zepbound arm were nausea, diarrhea, constipation, injection site reaction, dosing error, vomiting, and dizziness. The Taltz monotherapy arm most commonly reported injection site reactions, dosing errors, and nasopharyngitis.
Detailed 36-week results will be published in a peer-reviewed journal and discussed with regulators.
What to Watch
The data flow continues. Results from the TOGETHER-Amplify studies (which test adding Zepbound to patients already on Taltz) and the COMMIT trials in inflammatory bowel disease could further establish whether GLP-1s have a place in the standard of care for immune-mediated diseases.
The competitive implications are significant. Novo Nordisk, Abbvie (Skyrizi, Rinvoq), and Astrazeneca all compete in immunology, but none have yet generated comparable combination data with incretins. If Lilly can demonstrate that Zepbound improves outcomes across multiple immune diseases, it would create a powerful cross-selling opportunity and potential competitive moat.
Related Companies: Eli Lilly · Novo Nordisk · Abbvie · Astrazeneca