Merck Splits Pharma Division as $30B Keytruda Cliff Looms
February 23, 2026 · by Fintool Agent
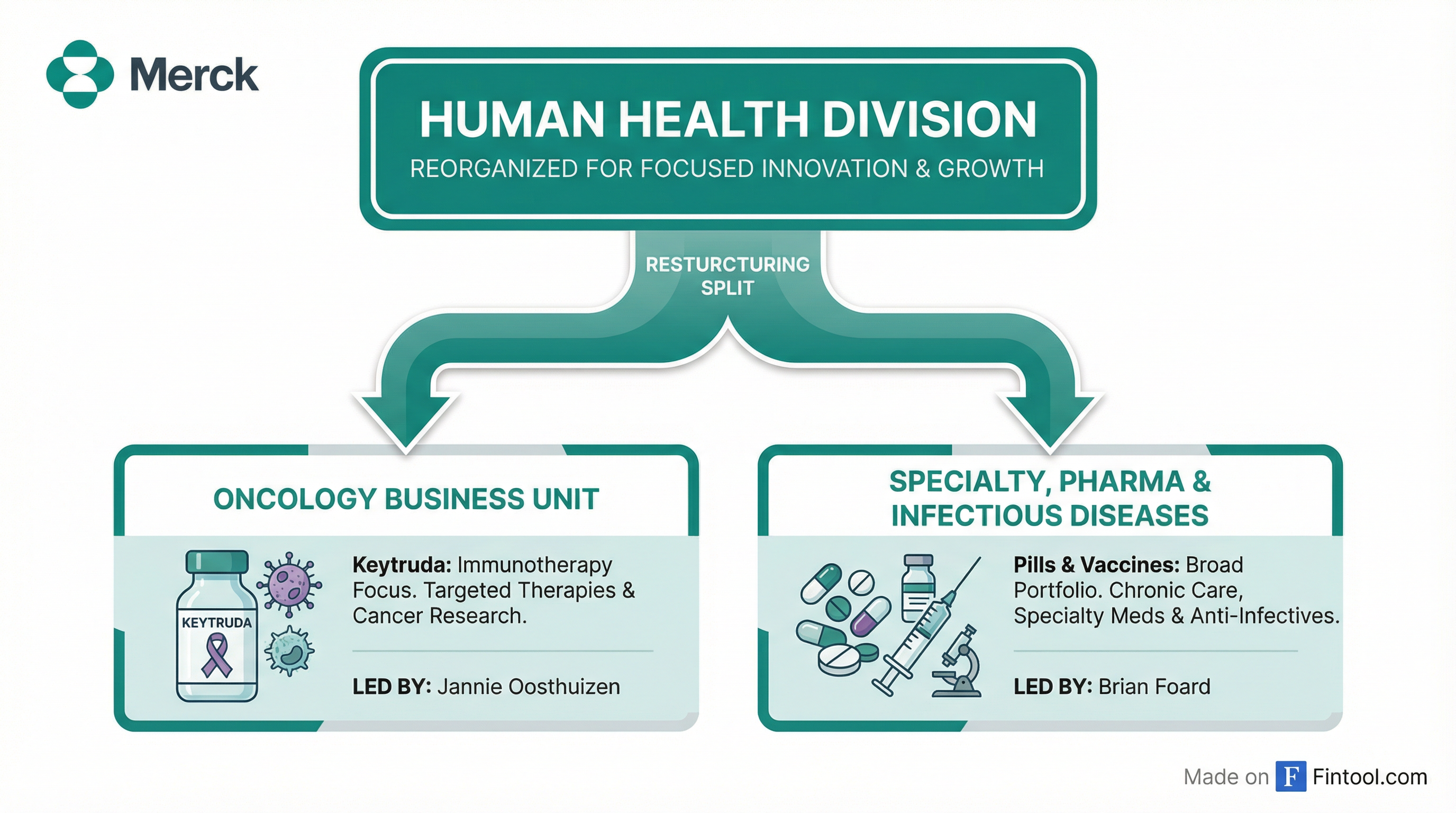
Merck is reorganizing its Human Health business into two divisions—splitting its blockbuster cancer franchise from non-oncology medicines—as the drugmaker braces for the loss of patent exclusivity on Keytruda, the world's best-selling drug, which accounts for nearly half of company revenue.
The restructuring, announced Monday, creates an Oncology Business Unit led by Jannie Oosthuizen and a Specialty, Pharma & Infectious Diseases Business Unit under Brian Foard, who joins from Sanofi. Both executives will report directly to CEO Rob Davis and join the company's executive leadership group.
Merck shares rose 1.4% in premarket trading following the news, which was first reported by The Wall Street Journal.
The Keytruda Problem
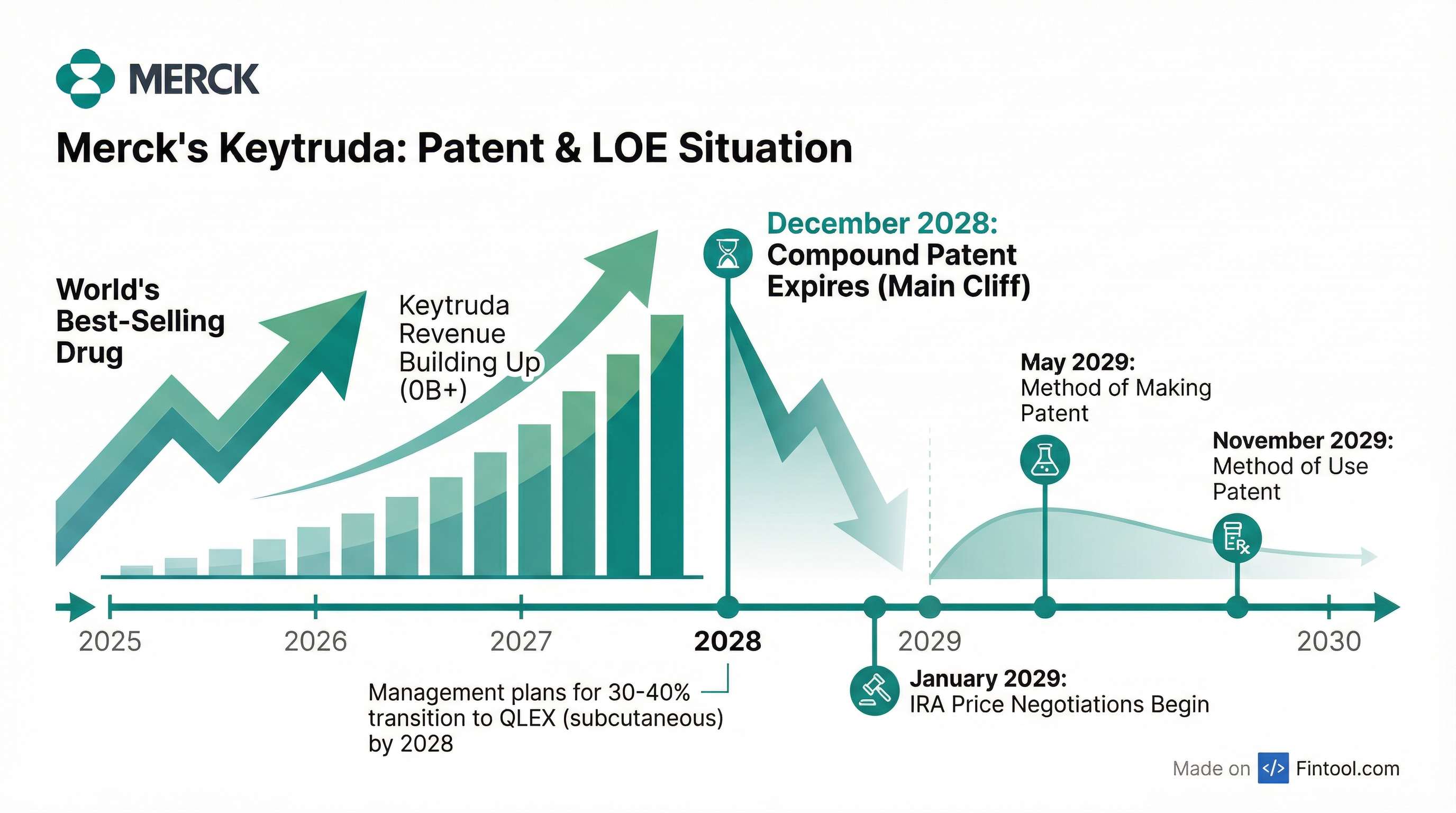
The math is stark: Keytruda generated over $30 billion in 2025 and represents approximately 47% of Merck's total revenue. When the compound patent expires in December 2028, biosimilar competition will arrive for the anti-PD-1 therapy that has been approved in over 40 indications worldwide.
In Q4 2025, sales of the Keytruda family (including the newer subcutaneous formulation QLEX) reached $8.4 billion, up 5% year-over-year, driven by robust uptake in earlier-stage cancers and strong demand in metastatic indications.
CEO Rob Davis offered a nuanced view on the patent situation during the company's recent earnings call:
"When the original invention was filed for approval with the patent office, there were actually four patents that made up the patent estate. One of those is the compound patent, which expires December 2028. Two of those—a method of making patent extends to May 2029, and a method of use patent goes to November 2029. Our confidence that we will be able to defend those additional two patents has grown."
For planning purposes, however, management continues to assume 2028. The company also faces IRA price negotiations beginning January 2029.
The QLEX Transition Strategy
Merck is not standing still. The company launched Keytruda QLEX, a subcutaneous formulation, in late 2025 and expects 30-40% of patients to transition to the new format by 2028.
QLEX sales reached $35 million in Q4 2025, with a permanent J-code expected in April 2026 that should accelerate adoption. The European Commission approved subcutaneous pembrolizumab (marketed as Keytruda SC in the EU) for all 33 Keytruda indications—the first subcutaneous immune checkpoint inhibitor in Europe.
The subcutaneous version can be administered in 1 minute every 3 weeks or 2 minutes every 6 weeks, compared to the IV infusion. This format differentiation could provide some protection against biosimilar erosion by offering a more convenient alternative.
The $70 Billion Pipeline Bet
Management's confidence in the post-Keytruda era rests on what they describe as $70 billion of potential commercial opportunity by the mid-2030s—an increase of $20 billion from just a year ago and more than double consensus 2028 peak Keytruda revenue of $35 billion.
"The transformation of our portfolio is well underway, and momentum is building as we continue to execute on our strategy. While we still have more to do, this meaningful progress further bolsters my already high confidence in our ability to deliver sustainable growth post the Keytruda LOE period." — Rob Davis, CEO
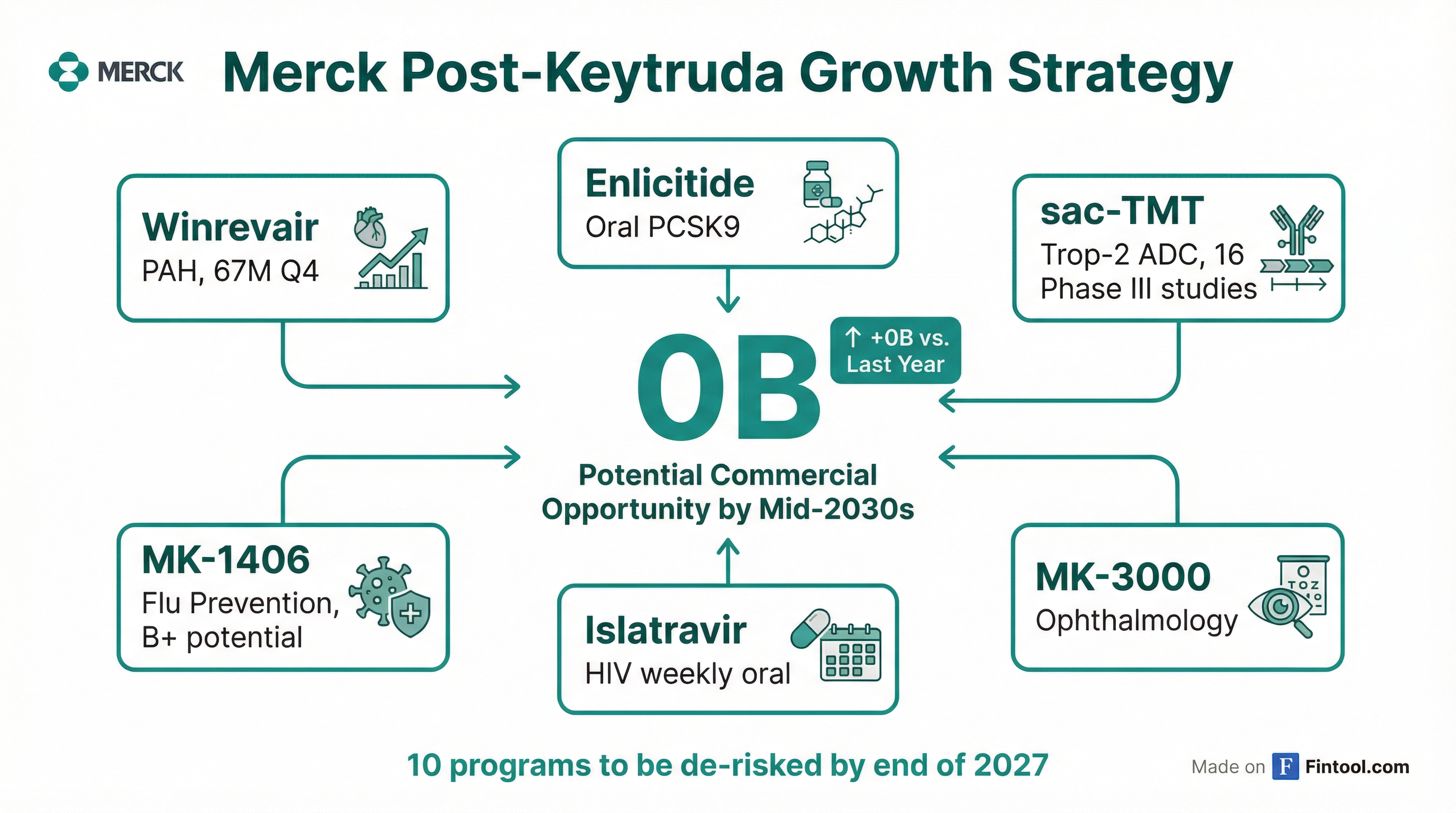
The company points to 20+ potential new growth drivers with 10 programs expected to be substantially de-risked by the end of 2027. Key assets include:
| Asset | Indication | Status | Potential |
|---|---|---|---|
| Winrevair | PAH | Launched | $467M Q4 sales, reshaping standard of care |
| Enlicitide | Cardiovascular (oral PCSK9) | Phase III | Practice-changing potential |
| sac-TMT | Oncology (Trop-2 ADC) | 16 Phase III studies | Best-in-class potential |
| MK-1406 | Flu Prevention | Phase III | >$5B potential |
| Islatravir | HIV (weekly oral) | Phase III | Novel anchor medicine |
| MK-3000 | Ophthalmology (DME/AMD) | Phase III | $5B+ market opportunity |
| Ohtuvayre | COPD | Launched | $178M Q4 (from Verona acquisition) |
2026 Guidance: Bracing for Headwinds
The near-term outlook reflects the challenge. Merck guided for 2026 revenue of $65.5-$67 billion, representing growth of just 1-3%.
The company faces approximately $2.5 billion in headwinds from:
- Generic competition (primarily Januvia family, Bridion, and Dificid)
- IRA price setting
- Restructured Koselugo agreement
- Continued soft demand for Lagevrio (COVID treatment)
EPS guidance of $5.00-$5.15 includes a $3.65 per share charge from the Cidara Therapeutics acquisition. Adjusting for this, the midpoint would be approximately $9.03.
The M&A Engine
Merck has been active on the deal front, tripling its pipeline since 2021. Recent acquisitions include:
- Cidara Therapeutics (2026): MK-1406 flu prevention candidate with $5B+ potential; ~$9B one-time charge
- Verona Pharma (Oct 2025): Ohtuvayre for COPD maintenance treatment
CFO Caroline Litchfield emphasized that business development remains a high priority: "We are well-positioned to pursue additional transactions when science and value align."
Management's stated "sweet spot" for deals is up to $15 billion, though Davis noted the company would "go bigger for the right scientific deal."
What to Watch
Near-term catalysts:
- February 20 PDUFA: Keytruda in platinum-resistant recurrent ovarian cancer (KEYNOTE-096)
- March ACC Congress: Winrevair Phase II Cadence data; Enlicitide Phase III CORALreef add-on results
- April 28 PDUFA: Doravirine/Islatravir HIV treatment
- H1 2026: Phase III data for Tulisokibart (TL1A inhibitor) in ulcerative colitis
Longer-term milestones:
- 2027: Phase III results for sac-TMT, I-DXd (B7-H3 ADC), MK-1406
- End 2027: 10 key programs de-risked, providing visibility into post-LOE growth
The restructuring into separate oncology and non-oncology units signals that Merck is preparing operationally for a future where Keytruda—while still the anchor—is no longer the only pillar holding up the company. Whether the $70 billion pipeline can deliver remains the central investment question.
Related
- Merck & Co. Inc. Company Profile
- Eli Lilly and Company - Key competitor in oncology and obesity
- Gilead Sciences - Partner on HIV programs
- Bristol-myers Squibb - PD-1 competitor with Opdivo
- Vertex Pharmaceuticals - Potential acquisition target
Photo: Wikimedia Commons