Earnings summaries and quarterly performance for AMGEN.
Executive leadership at AMGEN.
Robert A. Bradway
Chief Executive Officer and President
David M. Reese
Executive Vice President and Chief Technology Officer
James E. Bradner
Executive Vice President, Research and Development
Murdo Gordon
Executive Vice President, Global Commercial Operations
Peter H. Griffith
Executive Vice President and Chief Financial Officer
Board of directors at AMGEN.
Amy E. Miles
Director
Brian J. Druker
Director
Charles M. Holley, Jr.
Director
Ellen J. Kullman
Director
Greg C. Garland
Director
Mary E. Klotman
Director
Michael V. Drake
Director
Robert A. Eckert
Lead Independent Director
S. Omar Ishrak
Director
Tyler Jacks
Director
Wanda M. Austin
Director
Research analysts who have asked questions during AMGEN earnings calls.
Salveen Richter
Goldman Sachs
8 questions for AMGN
Terence Flynn
Morgan Stanley
8 questions for AMGN
Yaron Werber
TD Cowen
8 questions for AMGN
David Amsellem
Piper Sandler Companies
7 questions for AMGN
Christopher Schott
JPMorgan Chase & Co.
6 questions for AMGN
Jay Olson
Oppenheimer & Co. Inc.
6 questions for AMGN
Courtney Breen
AllianceBernstein
5 questions for AMGN
Evan Seigerman
BMO Capital Markets
5 questions for AMGN
Matthew Phipps
William Blair
5 questions for AMGN
Umer Raffat
Evercore ISI
5 questions for AMGN
David Risinger
Leerink Partners
4 questions for AMGN
Michael Yee
Jefferies
4 questions for AMGN
Mohit Bansal
Wells Fargo & Company
4 questions for AMGN
Alexandria Hammond
Wolfe Research
3 questions for AMGN
Gregory Renza
RBC Capital Markets
3 questions for AMGN
Alex Hammond
Sidoti & Company, LLC
2 questions for AMGN
Carter L. Gould
Barclays
2 questions for AMGN
Chris Schott
JPMorgan Chase & Company
2 questions for AMGN
Louise Chen
Cantor Fitzgerald
2 questions for AMGN
Christopher Raymond
Piper Sandler
1 question for AMGN
Conor MacKay
BMO Capital Markets
1 question for AMGN
Geoff Meacham
Citigroup Inc.
1 question for AMGN
James Shin
Analyst
1 question for AMGN
Luca Issi
RBC Capital Markets
1 question for AMGN
Michael DiFiore
Evercore ISI
1 question for AMGN
Michael Gee
UBS Financial Services Inc.
1 question for AMGN
Mike DiFiore
Evercore ISI
1 question for AMGN
Olivia Brayer
Cantor
1 question for AMGN
Sadia Rahman
Wells Fargo
1 question for AMGN
Timothy Anderson
BofA Securities
1 question for AMGN
Trung Huynh
UBS Group AG
1 question for AMGN
Recent press releases and 8-K filings for AMGN.
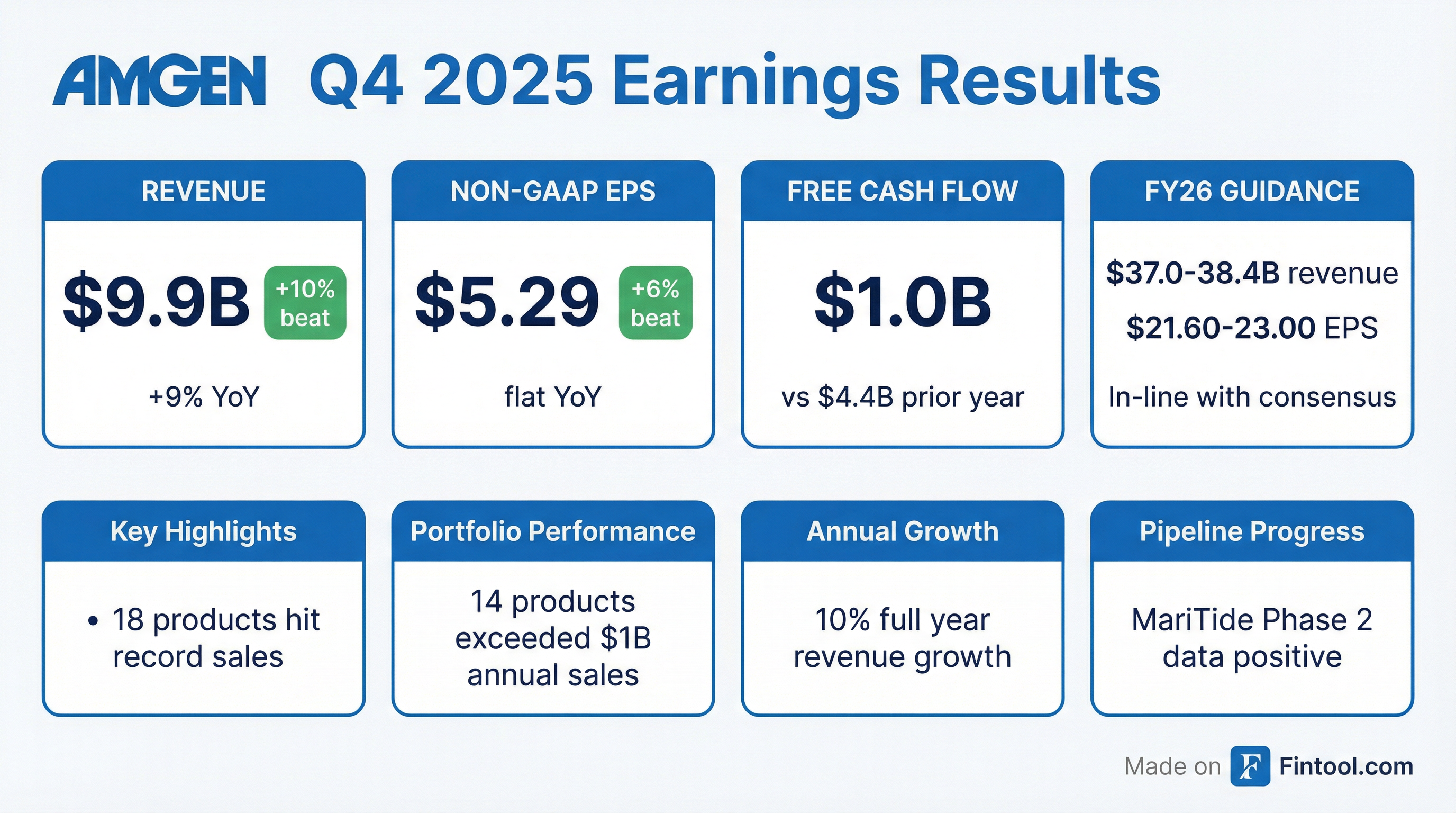
- Amgen closed 2025 with 13 products delivering double-digit growth, 14 products exceeding $1 billion in annual sales, and 18 records, driving double-digit revenue and EPS growth. Key drivers included Repatha, Evenity, Tezspire (each +30%+), a $5 billion rare-disease portfolio (+14%), and a $3 billion biosimilars portfolio (+37%) in 2025.
- The 2026 pipeline features six global phase III studies, led by MariTide (monthly to quarterly dosing for obesity/diabetes), the Olpasiran OCEAN(a) outcome study (timing delayed), and two phase III Sjögren’s studies of Dazodalibep (completion expected H2 2026).
- Expect a typical Q1 seasonal headwind from U.S. insurance cycles, lower Otezla/IMRALDI sales, €282 million of 2025 European Otezla sales facing generic entry, Prolia/XGEVA biosimilar erosion, and $250 million of Q4 2025 inventory build; Q1 non-GAAP operating margin guided at ~43%, lowest in the year.
- Having restored its balance sheet post-Horizon integration, Amgen maintains disciplined inorganic strategy—prioritizing best innovation, cash-on-cash returns, existing research synergies, and rapid integration.
- Amgen closed 2025 with broad momentum: 13 products achieved double-digit growth, 14 exceeded $1 billion in annual sales, its rare disease portfolio reached $5 billion (+14% y-o-y), and flagship franchises Repatha, Evenity, and Tezspire each grew >30% y-o-y【1】.
- For Q1 2026, Amgen anticipates typical seasonal headwinds—including U.S. insurance cycle impacts, Otezla facing EU generic entry (2025 EU sales €282 million), ongoing Prolia/XGEVA biosimilar erosion, and a $250 million inventory build—and expects non-GAAP operating margin near 43% (Q4 2025 level)【2】.
- The 2026 pipeline features 6 global Phase 3 studies of MariTide across weight management, Type 2 diabetes, obstructive sleep apnea, heart failure, and ASCVD (with data readouts into next year); an event-driven OCEAN(a) trial of Olpasiran has been delayed; and two Phase 3 trials of dazodalibep in Sjögren’s disease complete in H2 2026【1】【2】.
- Having restored its committed leverage post-Horizon deal, Amgen remains structurally agnostic on M&A, prioritizing best-in-class innovation, accretive cash returns, existing research alignment, and rapid integration in any inorganic transaction【9】.
- Amgen closed 2025 with strong momentum: 13 products delivered double-digit growth, 14 products exceeded $1 billion in annual sales, and 18 products achieved record performance, supporting double-digit revenue and EPS growth in 2025.
- The company’s six key growth drivers are Repatha, Evenity, Tezspire, its rare disease portfolio ($5 billion in 2025 sales, +14%), innovative oncology BiTE platforms (e.g., IMDELLTRA, Xaluritamig), and biosimilars ($3 billion in 2025 sales, +37%).
- A robust Phase III pipeline includes MariTide (monthly to quarterly dosing for obesity and type 2 diabetes), the OCEAN(a) olpasiran cardiovascular outcome study, and two Phase III trials of dazodalibep in Sjögren’s disease (completion expected H2 2026).
- Amgen anticipates seasonal Q1 2026 headwinds from the US insurance cycle, lower Q1 sales for Otezla/Imraldi, accelerated erosion of Prolia/XGEVA, and a $250 million Q4 2025 inventory build, with non-GAAP operating margin around 43% in Q1 2026.
- With its balance sheet restored post-Horizon integration, Amgen remains open to M&A, licensing, and partnerships, focusing on innovation value, cash returns, research synergies, and rapid integration.
- On March 4, 2026, Amgen’s Board declared a $2.52 per share dividend for the second quarter of 2026.
- The dividend will be paid on June 5, 2026.
- Shareholders of record as of May 15, 2026 will receive the dividend.
- 2025 performance: 13 products with double-digit growth, 14 exceeding $1 billion in annual sales and 18 record performers, driving double-digit revenue and EPS growth.
- Key growth drivers in 2025: Repatha, EVENITY and TEZSPIRE each grew > 30% yoy; rare disease portfolio delivered $5 billion (+14%; UPLIZNA +73%); biosimilars generated $3 billion (+37%).
- 2026 Q1 headwinds: seasonal U.S. insurance cycle, Otezla EU generic entry (€282 million sales in 2025), Prolia/Xgeva biosimilar erosion and prior quarter inventory build; non-GAAP operating margin ~43%.
- Commercial initiative: Repatha AmgenNow direct-to-patient program at $239/month, with > 5,000 patients enrolled, alongside preferred formulary placement and reduced copays (< $50/month).
- Pipeline focus: MariTide (monthly dosing for obesity/diabetes), olpasiran Lp(a) OCEAN(a) outcome study progressing, dazodalibep Sjögren’s phase III readout expected H2 2026.
- 2025 performance summary: 13 products delivered double-digit growth, 14 exceeded $1 billion in annual sales, and 18 produced record performance, driving double-digit revenue and EPS growth in 2025.
- Six key growth drivers delivered momentum: Repatha, EVENITY, and TEZSPIRE each grew over 30% and became multi-billion dollar franchises; the rare disease portfolio reached $5 billion (+14%); biosimilars generated $3 billion (+37%).
- Pipeline highlights include MariTide’s potential for monthly or less frequent obesity dosing, the OCEAN(a) olpasiran outcome study progressing with a later completion date, and fully enrolled phase III trials of dazodalibep in Sjögren’s disease set to complete in H2 2026.
- Q1 headwinds expected from U.S. insurance benefit cycles, higher co-pays, European Otezla generic entry ($282 million 2025 sales), biosimilar erosion of Prolia/Xgeva, and a $250 million inventory build in Q4 2025; Q1 non-GAAP operating margin forecast to mirror Q4 2025 at ~43%.
- Exited 2025 with broad momentum: 13 products delivered double-digit growth, 14 exceeded $1 billion in annual sales, and 18 achieved record performance, driving double-digit revenue and EPS growth.
- Q1 2026 expected to face seasonal U.S. insurance headwinds, lower Otezla/Enbrel sales, European generic entry for Otezla (€282 million 2025 sales) and accelerated Prolia/Xgeva erosion; non-GAAP operating margin guided at ~43%.
- Repatha access expanded: now preferred on virtually all U.S. formularies, average copay < $50/month, over 50% of Medicare patients with no prior authorization, plus AmgenNow direct-to-patient program at $239/month with > 5,000 enrolled.
- Uplizna uptake in IgG4-related disease: ~500 unique prescribers treating a 35,000-patient market across all therapy lines; myasthenia gravis launch split 50% bio-naive/bio-experienced prescriptions.
- Pipeline focus on differentiated assets: MariTide for obesity with monthly dosing, multiple phase II/III studies underway; olpasiran’s event-driven OCEAN(a) trial delayed; dazodalibep Sjögren’s phase III readouts expected H2 2026.
- On February 17, 2026, Amgen sold four tranches of senior unsecured notes totaling $4.0 billion, comprising $1.0 billion of 4.200% notes due 2031, $1.75 billion of 4.850% notes due 2036, $0.5 billion of 5.500% notes due 2046 and $0.75 billion of 5.650% notes due 2056, generating net proceeds of approximately $3.961495 billion.
- The notes pay interest semi-annually in arrears each February 19 and August 19, rank pari passu with all existing and future senior unsecured debt and are effectively subordinated to any secured obligations of Amgen’s subsidiaries.
- Holders have an optional put on a change-of-control event at 101% of principal plus accrued interest, and the company may optionally redeem notes at a make-whole premium prior to specified call dates.
- Amgen reaffirmed its oncology R&D framework to focus on transformative therapies in both hard-to-treat solid tumors and hematologic malignancies, anchored by its T-cell engager (BiTE) and precision small-molecule platforms.
- IMDELLTRA (DLL3 BiTE) has full U.S. approval in second-line small cell lung cancer, is administered at over 1,600 U.S. sites, and is being evaluated in three additional Phase 3 trials (first-line metastatic, maintenance and limited stage), plus subcutaneous and extended-interval dosing studies to improve access.
- BLINCYTO’s development includes a subcutaneous formulation and earlier-line B-ALL trials (e.g., Golden Gate in patients ≥55 years), with Phase II studies underway in SLE and refractory rheumatoid arthritis, aiming to enhance convenience and expand indications.
- Xaluritamig (STEAP1 BiTE) is advancing in Phase III for post-taxane and pre-taxane metastatic castration-resistant prostate cancer—designed to demonstrate overall survival benefit versus chemotherapy without requiring biomarker gating—and is also being explored in Ewing sarcoma.
- Amgen reaffirmed its R&D framework targeting differentiated, transformative therapies across hard-to-treat solid tumors and select hematologic malignancies, with two core modalities—T-cell engagers and precision small molecules—and selective ADC investments leveraging its biologics and chemistry strengths.
- The DLL3-targeting BiTE IMDELLTRA received full approval in second-line or later small cell lung cancer, now administered at over 1,600 U.S. sites (mostly community); three pivotal phase III trials in first-line metastatic, maintenance and limited-stage settings are ongoing globally.
- KRAS G12C inhibitor LUMAKRAS is approved in second-line NSCLC and third-line colorectal cancer; ongoing studies include CodeBreaK 202 (chemotherapy ± LUMAKRAS vs. chemo + pembrolizumab in first-line NSCLC) and a first-line CRC trial of LUMAKRAS + Vectibix + FOLFIRI, which showed ~55% ORR and >90% DCR in phase II.
- STEAP1-targeting BiTE xaluritamig is being advanced in prostate cancer with survival-driven pivotal trials both pre- and post-taxane (chemo-free in the former) and in relapsed/refractory Ewing sarcoma, aiming to fill unmet need without biomarker gating.
Fintool News
In-depth analysis and coverage of AMGEN.
Quarterly earnings call transcripts for AMGEN.
Ask Fintool AI Agent
Get instant answers from SEC filings, earnings calls & more